Original Article – DOI: 10.33594/000000857
Accepted 19.03.2026 - Published online 30.03.2026
Cellular Physiology and Biochemistry (60): 127 - 135
Lactic Fermentation in Traditional Beverages Made from Cereals and Tubers. Process Control Using a Biosensor
bFacultad de Ciencias de la Salud, Universidad Técnica de Manabí, Portoviejo,Ecuador,
cDepartamento de Posgrado, Facultad de Posgrado, Universidad Técnica de Manabí, Ecuador,
dDepartamento de Procesos Químicos, Alimentos yBiotecnología, Grupo de Investigación: Seguridad Conservación e Innovación de Alimentos y Procesos, Facultad de Ingeniería y Ciencias Aplicadas. Universidad Técnica de Manabí, Portoviejo, Ecuador,
eDepartamento de Ingeniería Industrial, Facultad de Ingeniería y Ciencias Aplicadas, Universidad Técnica de Manabí, Portoviejo, Ecuador,
fCarrera de Agroindustria, Escuela Superior Politécnica Agropecuaria de Manabí Manuel Félix López, ESPAM–MFL, Calceta 130250, Ecuador
Keywords
Abstract
Background/Aims: Monitoring lactic acid is a critical parameter during the production of fermented foods, as it serves as an indicator of product quality and process control. The use of biosensors offers a simple and cost-effective approach for real-time monitoring in the production chain. Methods: A biosensor based on lactate oxidase (LacOx) was developed to determine lactic acid content in fermented beverages (chicha and pozol). Different membranes were evaluated as immobilization supports, with particular focus on enzyme stability. The system operated at a potential of –0.5 mV, and the amperometric signal was recorded after 10 seconds. Sensor performance was assessed through calibration, sensitivity, and kinetic analysis. Results: The enzymatic reaction rate was directly proportional to oxygen consumption (mg L⁻¹ O₂ s⁻¹), showing a linear detection range between 0.1 and 2 mM (R² = 0.9986). The sensor demonstrated good sensitivity at 0.1 mM and allowed membrane reuse up to 15 times. Enhanced stability was observed when using a cassava biopolymer as immobilization support. The Michaelis–Menten constant (KM = 340 µM) indicated a high affinity of the enzyme for the substrate. Conclusion: The developed biosensor provides a reliable, sensitive, and low-cost method for monitoring lactic acid in fermented beverages and represents a practical tool for quality control in the food industry.
Introduction
The importance of fermented foods in cultures around the world continues to grow, with an estimated 5,000 different types of fermented foods being consumed [1]. In the case of fermented beverages, cereals and tubers undergo biochemical transformations, becoming ideal substrates for fermentation and promoting the growth of microorganisms capable of transforming sugars into alcohol and lactic acid. Sugars and starches are metabolized to form the compounds of interest. The biochemical process for the fermentation of cereals usually begins by soaking the grains for several days. During this soaking, various microorganisms form a population that becomes dominated by lactic acid bacteria (LAB). The main source of energy for the microorganisms is the fermentable sugars generated by the amylases present in the cereals. Through this mechanism, lactic acid and other components are obtained at the end of the process. The effects of this process can be described in terms of preservation and increased functional properties. In the first case, the different organic acids that are formed allow for the biopreservation of beverages, since the final pH causes the bacterial cytoplasmic membrane to rupture, inhibiting active transport and altering its basic functions. Thus, functional properties are increased because the bioactive compounds present in cereals, such as dietary fibers and polyphenols, increase their bioavailability, improving the antioxidant potential and probiotic content of the final product [2]. In general, lactic fermentation allows for preservation, improves the texture and mouthfeel of foods, while its functional properties provide health benefits. Recent studies have reported that high concentrations of lactic acid and nutraceuticals present in fermented beverages can promote liver recovery (liver damage) caused by ethanol, as it restores antioxidant and phytochemical levels, suppressing liver inflammation and thus significantly improving metabolic disorders [3]. Other studies have reported the nutritional benefits of lactic bacterial fermentation in beverages due to the production of γ-aminobutyric acid, which is an inhibitor responsible for reducing depression and anxiety [4]. In this context, quality control in fermented beverages using a biosensor could be useful for the food industry, as it would allow the presence of lactic acid to be monitored in parallel with the formation of ethanol and other active compounds, facilitating its management during the fermentation process.
L-lactate + LacOx pyruvate + LacOx red
The detection mechanism is based on the enzymatic oxidation of L-lactate catalyzed by lactate oxidase, in which oxygen is consumed and hydrogen peroxide is produced (Fig. 1). The resulting change in oxygen concentration (or the generated hydrogen peroxide) is detected electrochemically and is proportional to the lactate concentration.
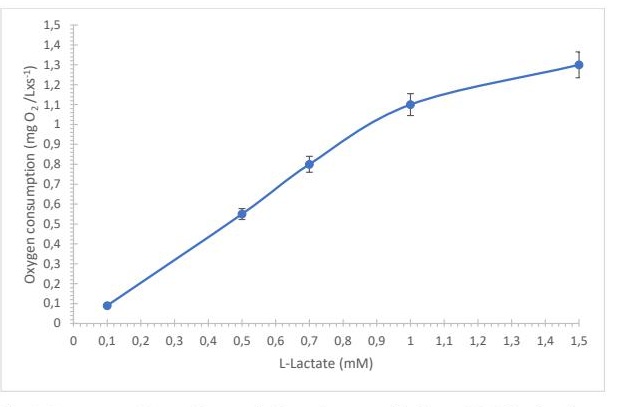
Fig. 1: Fig. 1. Response of the equipment during substrate oxidation at 35°C in phosphate buffer 0.05 M, pH 7.5.
LacOx red + O₂ LOx ox + H₂O₂ (eq. 1)
There are different methods for quantifying lactic acid in fermented beverages. For example, high-performance liquid chromatography was used to estimate the concentration of this organic acid in industrial yogurt production [5]. In another study, a colorimetric-enzymatic method was developed that uses boric acid as an inhibiting agent and as a substrate. The reaction is catalyzed by tyrosinase, which causes a color change proportional to the concentration of lactic acid [6]. Methods for high-precision aqueous solutions have also been developed by determining titratable acidity using potentiometers. It was reported that reducing sugars do not interfere with the measurements [7]. In craft beer, the presence of lactic acid as a marker of quality and indicator of the absence of pathogenic bacteria such as salmonella has been made possible through the use of a biosensor [8]. Thus, the objective of this study was to develop a biosensor for quality control in the beverage industry to monitor lactic acid formation in a simple and low-cost manner in products made from cereals and tubers.
Materials and Methods
Chemicals and reagents LacOx E.C.232 -841-6 from Pediococcus sp, with each vial containing 2.9 mg of protein, L-lactate, glutaraldehyde (50%), L-(+) lactic acid, HPLC grade sulfuric acid, and Flavin adenine dinucleotide were from Sigma (Sigma-Aldrich, St Louis, MO, USA). The preactivated Immunodyne ABC membrane (Nylon 6, 6 pore size 0.45 µm) was supplied by Pall Europe (Portsmouth, UK). Sodium phosphate, sodium acetate anhydrous, was from Panreac (Panreac Química, Barcelona, Spain). Bidistilled milli-Q water was used throughout.
Preparation of the LacOx solution The commercial enzyme was dissolved in 5 mL of 10mM buffer solution at pH 7.5, with an activity of 10.2 U/mL aliquoted in 0.5 mL Eppendorf tubes and frozen stored at -30 ºC until use.
Equipment The sensor used in this study is constructed with a platinum electrode, model 20 Dual Digital Model (Rank Brothers, Bottisham, Cambridge, England), in which an immobilized enzyme was used. This electrode consists of two internal sections: 1) a working electrode with a 2 mm diameter platinum disc, and 2) a reference electrode with a surrounding silver ring (Ag/AgCl). The instrument has a thermostatic reaction cell and a preactivated oxygen-permeable immunodine ABC membrane. Chromatographic analysis was performed using a YL9100 system manufactured by Young Lin Instrument CO., LTD (Korea).
Sample preparation To avoid interference with ethanol, it was necessary to dilute the chicha (made from cassava) and pozol (made from corn) to approximately 1 or 2 degrees of alcohol in phosphate buffer solution (pH 7.1). The samples were then filtered using filter paper (0.45 µm) to remove any suspended particles. The concentration of lactic acid in the samples was determined using a calibration graph obtained with standard lactic acid solutions.
Biosensor preparation and mode of operation The sensor's operating method was previously described [12, 14]. The oxidation of L-lactate takes place at the platinum electrode. To start the process, the chamber must be saturated with 100% oxygen in order to establish the sensitivity of the instrument. This step consists of filling the reaction cell with distilled water saturated with oxygen while stirring. The entire process takes less than 10 seconds. The reaction cell is maintained at the optimum temperature for the enzyme (35°C) by recirculating water, which has been previously heated with a thermostat. To separate the enzyme solution (inside the reaction cell) from the electrode base, the Inmmunodyne ABC membrane is used, which isolates the electrode and only allows oxygen to pass through.
Immobilized enzyme sensor The appropriate percentage of glutaraldehyde for the enzyme (LacOx) was studied in a range between 0.5% and 3% and set at 1.5%. Consequently, the total mixture (50 µL) contained 15 µL of LacOx solution (10.2 U/mL), 29 µL of 0.5 M sodium phosphate buffer, pH 7.0, and 6 µL of glutaraldehyde. It was then deposited on the Immuno-Dyne ABC membrane (1 cm²) and left to dry for one hour before starting the immobilization process. After drying, 5 µL of 2% cellulose acetate solution in acetone was added to prevent any matrix interference [13]. The two membranes (LacOx and Teflon) were then attached to the oxygen electrode using a rubber ring and placed in the thermostatic reaction cell.
Description of the chromatographic method To determine the presence of lactic acid in fermented beverages or standards, we used a Rezex ROA-Organic Acid H+ thermostatic column (8 µm, x300 x7.8 mm) at 45°C, together with sulfuric acid at a concentration of 0.095 N. We used 15 µL of the sample diluted with sulfuric acid and a flow rate of 1 mL/min in the mobile phase with the help of a peristaltic pump. Fluorescence detection was used at excitation and emission wavelengths of 230 and 450 nm, respectively. The gradient was linear, and two solvents were used: 20 mM sodium acetate buffer, pH 7.2 (solvent A), and 100 mM sodium acetate pH 7.2/acetonitrile/methanol (30:35:35) v/v as solvent B. The mobile phase was passed through a 0.45 µm nylon filter and then degassed. Quantification was performed using a calibration curve between 0.1 mM and 2 mM lactic acid.
Application of the enzyme sensor to analyze fermented beverages The enzyme sensor was used to quantify lactic acid in chicha (made from cassava) and pozol (made from corn). The method was validated and its results were compared with those obtained using HPLC.
Validation tests The reproducibility of the system was evaluated by injecting 30 consecutive standards of 1 mM lactic acid or fermented beverage samples into the immobilized enzyme system on the same day, using the same equipment and under the same conditions. Linearity was achieved by analyzing three times the standards ranging from 0.1 to 1 mM for the immobilized enzyme system using the ordinary least squares (OLS) regression method. The sample dilution was 1/10 to obtain a strong signal, avoiding interference from the matrices. It was necessary to determine the stability of the membrane under operating conditions. To do this, successive injections of 0.5 mM lactic acid were applied to a single membrane and the response was recorded until 50% of its initial activity was observed. The results were expressed as a function of time. The correlation between the sensor and HPLC was studied using different concentrations.
Statistical methods The increase in standard concentrations is analyzed using Statgraphics Plus software (v5.1). The linearity of the data between the sensor and the HPLC is determined by the coefficient of determination (R²) and the standard error (SE). A two-sample t-test (two-tailed) is used to determine if the HPLC and biosensor are statistically equivalent. The null hypothesis states that the population means are the same (H0: µ sensor = µ HPLC) while the alternative hypothesis states that the means are not equal (H0: µ sensor ≠ µ HPLC).
Results
Biosensor optimization The consumption of oxygen during the enzymatic reaction (eq. 1) allowed the determination of lactic acid concentration in the analyzed samples. The operating conditions of the enzyme were evaluated, and the best response was achieved at pH 7.5 and 35°C (Fig. 2). The system showed minimal background noise at 4 seconds when applying a negative potential of –0.5 mV (tested range: –0.1 to –1.5 mV). The signal obtained in the transducer was amplified and recorded for further processing (Fig. 3). Dispersion, expressed as coefficient of variation (CV), was evaluated in fermented beverages (chicha and pozol) and standard solutions. The CV values were 1.3%, 1.1%, and 1.5%, respectively. The variation between consecutive injections of a 0.5 mM standard or fermented beverage samples was determined under identical conditions. The values for corn-based beverages, cassava-based beverages, and the standard were 1.3%, 2.3%, and 3.5%, respectively.
Fig. 2: Schematic representation of key principle of developing a biosensor.
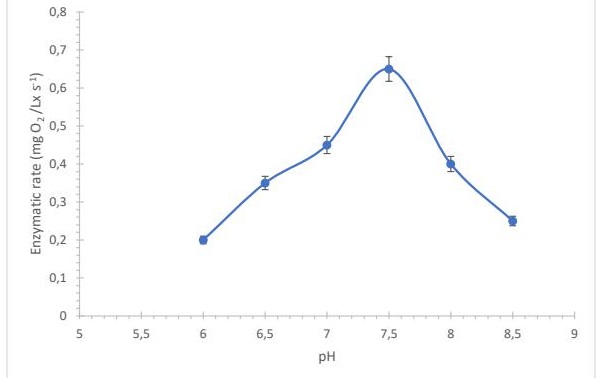
Fig. 3: Effect of pH on the reaction rate of the enzyme lactate oxidase determined by the enzyme sensor. n=3.
Kinetic parameters The Michaelis–Menten constant (KM) was determined to evaluate the affinity of the enzyme for the substrate. In the free state, the KM value was 340 µM. When different immobilization membranes were tested, a decrease in enzyme affinity was observed. A reduction of up to 22% was found using the Immuno-Dyne ABC membrane, whereas a reduction of 10% was observed when using a cassava-based biopolymer. The enzymatic reaction followed Michaelis–Menten kinetics, described by:
v = (Vmax × [S]) / (KM + [S]) (eq. 2)
The detection was linear for lactic acid concentrations in the range of 0.1–2 mM (Fig. 4), with a coefficient of determination of R² = 0.9986. The detection limit was 0.1 mM. The enzyme–substrate relationship was further analyzed using:
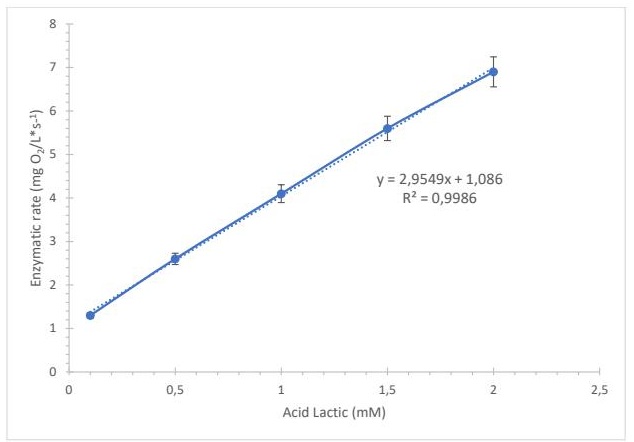
Fig. 4: Calibration curve obtained by the sensor with the immobilised enzyme. n=3.
[S]/v = [S]/Vmax + KM/Vmax (eq. 3)
Stability of enzymes using different supports The stability of LacOx was evaluated using Immuno-Dyne ABC and cassava-based biopolymer membranes at 4°C. The cassava-based membrane maintained 90% of its initial activity after 75 days, while the Immuno-Dyne ABC membrane reached the same level after 55 days (Fig. 5). The membranes could be reused up to 15 times without significant loss of activity.
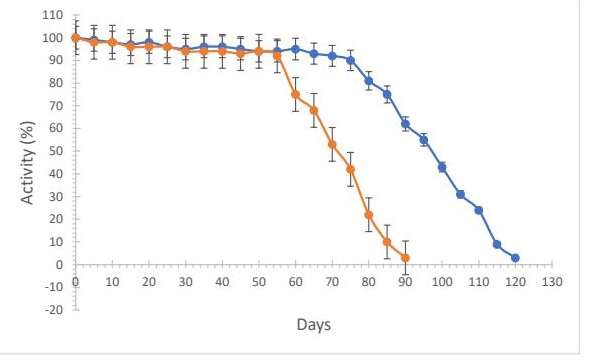
Fig. 5: Stability of immobilized LacOX stored under refrigeration. Blue line with cassava biopolymer. Orange line with Immuno-Dyne ABC.
Comparison with HPLC A strong correlation between the biosensor and HPLC measurements was observed (R² = 0.9966) (Fig. 6). A two-sample t-test showed no significant difference between biosensor (Ẋ = 1.4; s = 0.1216) and HPLC (Ẋ = 1.16; s = 0.1252) measurements (t = 0.0553; p = 0.954; df = 30), confirming good agreement between both methods (Table 1). The variability of the biosensor reached up to 3.4%, while HPLC variability did not exceed 1.7%.
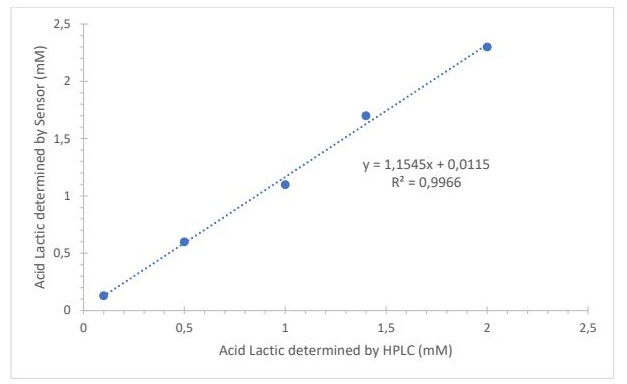
Fig. 6: Correlation between the lactic acid content in fermented beverages (chicha) obtained with the sensor and HPLC. n=3.
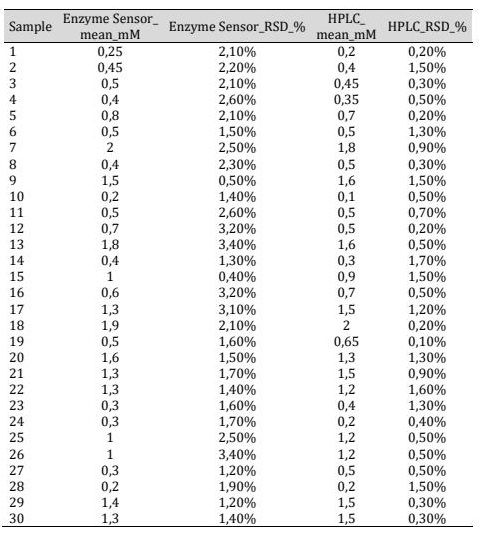
Table 1: Measurements lactic acid levels by the enzime sensor and HPLC. Results are expressed in mM (n=3).
Discussion
The results demonstrate that the developed biosensor provides a reliable and reproducible method for quantifying lactic acid in fermented beverages. The low coefficient of variation indicates high precision and stability of the system. The selected operating conditions (pH 7.5 and 35°C) are consistent with optimal activity ranges reported for lactate oxidase, supporting the validity of the experimental setup. The observed decrease in enzyme affinity after immobilization is expected, as structural constraints and limited substrate accessibility typically affect enzyme performance. However, the relatively small decrease observed with the cassava-based biopolymer indicates that this support provides a more favorable microenvironment compared to the Immuno-Dyne ABC membrane. The linear detection range (0.1–2 mM) and high coefficient of determination (R² = 0.9986) confirm the robustness of the biosensor and are consistent with previously reported systems [9–11]. The high stability of the cassava-based membrane, maintaining 90% activity after 75 days, suggests that this material offers improved enzyme immobilization and protection. This behavior is in agreement with previous findings [12] and may be attributed to the larger surface area and enhanced binding capacity of the biopolymer. The strong correlation with HPLC results and the absence of significant differences between methods confirm the analytical reliability of the biosensor. Although slightly higher variability was observed compared to HPLC, this can be attributed to factors such as membrane replacement and background signal fluctuations. Overall, the biosensor represents a practical alternative to conventional analytical methods, offering advantages in terms of simplicity, cost, and rapid response, making it suitable for real-time monitoring in food production processes.
Conclusion
The results indicate that the sensor was able to monitor the concentration of lactic acid in fermented beverages, with a detection limit (DL) of 0.1 mM, with low interference and good stability. The support that allowed the stability of the LacOx enzyme to be extended was the cassava biopolymer, which enabled a reduction in costs. The results were comparable to those obtained with high-performance liquid chromatography (HPLC). In conclusion, the method is a useful tool for the food industry due to its ease of use and low cost (one-fifth that of liquid chromatography).
Disclosure Statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Tamang, J. P., Holzapfel, W. H., Shin, D. H., & Felis, G. E. (2017). Editorial: Microbiology of ethnic fermented foods and alcoholic beverages of the world. Frontiers in Microbiology, 8, 1377. https://doi.org/10.3389/fmicb.2017.01377
- Arslan, S., Durak, A. N., Erbas, M., Tanriverdi, E., & Gulcan, U. (2015). Determination of microbiological and chemical properties of probiotic boza and its consumer acceptability. Journal of the American College of Nutrition, 34(1), 56-64. https://doi.org/10.1080/07315724.2014.880661
- Modi, R., Sahota, P., Singh, N., & Garg, M. (2023). Hepatoprotective and hypoglycemic effect of lactic acid fermented Indian gooseberry (Amla) beverage on chronic alcohol-induced liver damage and diabetes in rats. Food Hydrocolloids for Health, 4, 100155. https://doi.org/10.1016/j.fhfh.2023.100155
- Camillo, G., Bicas, J., & Junior, M. (2023). Nutritional benefits of fruit and vegetable beverages obtained by lactic acid fermentation. In Value-Addition in Beverages through Enzyme Technology (pp. 177-198). https://doi.org/10.1016/B978-0-323-85683-6.00002-8
- Gezginc, Y., Topcal, F., Comertpay, S., & Akyol, I. (2015). Quantitative analysis of the lactic acid and acetaldehyde produced by Streptococcus thermophilus and Lactobacillus bulgaricus strains isolated from traditional Turkish yogurts using HPLC. Journal of Dairy Science, 98(3), 1426-1434. https://doi.org/10.3168/jds.2014-8447
- Ran, Z., Wang, X., Zhang, L., Yang, Y., Shang, Z., Chen, Q., Ma, X., Qian, Z., & Liu, W. (2023). Enzymatic colorimetric method for turn-on determination of L-lactic acid through indicator displacement assay. Journal of Bioscience and Bioengineering, 136(3), 159-165. https://doi.org/10.1016/j.jbiosc.2023.06.001
- Feng, S., Xiang, S., Bian, X., & Li, G. (2020). Quantitative analysis of total acidity in aqueous lactic acid solutions by direct potentiometric titration. Microchemical Journal, 157, 105049. https://doi.org/10.1016/j.microc.2020.105049
- Jadán, F., Latorre, G., Sánchez, V., Piñero, A., Zambrano, M., Vivas, F., Gorozabel, A., Arteaga, A., Buchelli, L., Góngora, E., Zambrano, M., & Jadán, C. (2024). Development of a sensor to quantify lactic acid in beer. Arabian Journal of Chemistry, 17, 105639. https://doi.org/10.1016/j.arabjc.2024.105639
- Han, D., Li, X., Liang, Z., Zhao, B., Wu, Z., Han, F., Han, D., & Niu, L. (2023). Label-free photoelectric sensor for lactic acid determination in human sweat. Chinese Chemical Letters, 34, 107722. https://doi.org/10.1016/j.cclet.2022.08.002
- Zhao, Z., Zhu, J., & Xie, Y. (2017). Simple online pretreatment of column switching coupled with ion chromatography for the determination of lactic acid in lobaplatin. Journal of Pharmaceutical and Biomedical Analysis, 138, 316-321. https://doi.org/10.1016/j.jpba.2017.02.031
- Taleghani, N. I., Taleghani, N. A., & Yadav, V. (2025). A theoretical framework for the design and optimization of a novel point-of-care lactate biosensor. Next Research, 2, 101031. https://doi.org/10.1016/j.nexres.2025.101031
- Jadán, F., Villavicencio, C., Mieles, J., Cantos, M., Anchundia, A., Zambrano, M., Intriago, F., Giler, S., Pérez, E., Litardo, R., & Jadán, C. (2025). Quantification of lactic acid in wines using an amperometric biosensor. Food Control, 167, 110821. https://doi.org/10.1016/j.foodcont.2024.110821
- Qiong, C., Tuzhi, P., & Liju, Y. (1998). Silk fibroin/cellulose acetate membrane electrodes incorporating xanthine oxidase for the determination of fish freshness. Analytica Chimica Acta, 369, 245-251. https://doi.org/10.1016/S0003-2670(98)00213-X
- Vergara Velez, G., Real Pérez, G. L., Santana Moreira, G. J., Caballero Vera, H. H., Litardo Velásquez, R. M., Noriega Verdugo, D. D., Anchundia Loor, A. M., Arcentales Franco, C., Peña Agurto, M., Vera Delgado, J. M., Jadán Piedra, C., & Jadán Piedra, F. (2026). Selective quantification of tryptophan in milk using a sensor based on L-tryptophan dehydrogenase. Food Science and Biotechnology, 35(4), 1051-1058. https://doi.org/10.1007/s10068-026-02089-w https://doi.org/10.1007/s10068-026-02089-w