Original – DOI: 10.33594/000000860
Accepted 26.03.2026 - Published online 12.04.2026
Cellular Physiology and Biochemistry (60): 189 - 198
Characterization of a Biosensor For the Detection of Triglycerides in Vegetable Oils Using Lipase from Candida Rugosa
bDpto de Ingeniería Ambiental Agroalimentaria. Calidad y Seguridad y Salud en el Trabajo. Escuela Superior de
Ingeniería y Tecnología. Universidad Internacional de la Rioja (UNIR), Logroño, Spain,
cDepartamento de Posgrado, Facultad de Posgrado, Universidad Técnica de Manabí, Ecuador,
dDepartamento de Procesos Agroindustriales. Facultad de Agrociencias. Universidad Técnica de Manabí,
eDepartamento de Química, Facultad de Ciencias Básicas. Universidad Técnica de Manabí. Portoviejo, Ecuador,
fUniversidad Agraria del Ecuador. Guayaquil, Ecuador,
gUnidad Academica de Ciencias Agropecuarias. Universidad Católica de Cuenca. Campus la Troncal. Cañar, Ecuador,
hDepartamento de Ingenieria Industrial. Facultad de Ingeniería y Ciencias Aplicadas. Universidad Técnica de Manabí, Portoviejo, Ecuador,
iDepartamento de Procesos Agroindustriales. Facultad de Agrociencias. Universidad Técnica de Manabí,
jDepartamento de Procesos Químicos, Alimentos y Biotecnología, Grupo de Investigación: Seguridad Conservación e
Innovación de Alimentos y Procesos. Facultad de Ingeniería y Ciencias Aplicadas. Universidad Técnica de
Manabí, Portoviejo, Ecuador,
kDepartamento de Procesos Químicos, Alimentos y Biotecnología. Facultad de Ingeniería y Ciencias Aplicadas. Universidad Técnica de Manabí, Portoviejo, Ecuador
Keywords
Abstract
Background/Aims: Rapid and cost-effective quantification of triglycerides remains a challenge in industrial food quality control, where conventional analytical methods are often time-consuming and resource-intensive. This study aimed to develop and characterize an amperometric biosensor based on lipase (LPS) as a biorecognition element for efficient triglyceride detection in vegetable oils. Methods: Lipase was immobilized onto the electrode surface to catalyze the hydrolysis of glyceryl tributyrate, enabling electrochemical signal generation. Measurements were performed at an applied potential of −600 mV, with a detection time of 7 seconds. The enzymatic reaction rate was derived from the slope of the amperometric signal (mV/s), and calibration was conducted over a concentration range of 1–5 mM. Results: The biosensor demonstrated a strong linear response (R² = 0.998) and high substrate affinity (KM = 0.13 mM). The system exhibited excellent repeatability and specificity, with minimal interference from other triglycerides. Long-term stability studies showed that the immobilized enzyme retained approximately 90% of its activity over 260 days and could be reused up to 27 times without significant performance loss. Conclusion: This biosensor represents a robust, rapid, and environmentally friendly analytical platform for triglyceride detection, offering a practical alternative to conventional techniques and strong potential for implementation in routine industrial quality control.
Introduction
Medium-chain triglycerides (MCT) and short-chain triglycerides (SCT) are currently being studied for their potential applications in the food industry. They are produced by enzymatic transesterification because this process is highly efficient and considered fast, yielding values of up to 56.7% in fish oil (Zou et al., 2024; Zhang et al., 2024; Liu et al, 2024). Process control in the food industry is one of the main applications with the aim of achieving quality products, due to oxidation reactions which have an effect on the flavor and sensory profile of oils and enriched products. For example, it has been shown that lipase-catalyzed hydrolysis of butterfat accelerated the oxidation process, forming aromatic markers, and when applied to enriched milk, it improved its sensory profile (Xiang et al., 2025; Zhong et al., 2024). Another application of lipases in the food industry is their ability to modify the structure of polylactic acid (PLA) and produce lactic acid hydrolysates, with the aim of using biodegradable PLA as an environmentally friendly and sustainable alternative for food packaging using FDM 3D printing (Shi et al, 2025). The industrial use of lipases has spread as an efficient method to accelerate the kinetics of release and, consequently, the bioavailability of free fatty acids present in cheese, whose matrix encapsulates lipids, making them difficult to digest, thus becoming a solution to digestive disorders in older people (Liu et al, 2025). Thus, the development of this type of enzyme has been the subject of interest at the industrial level. This advance is possible through the technique of protein engineering and recombinant DNA technology, where lipases from recombinant microorganisms with specific characteristics in their kinetic properties, thermostability, greater substrate specificity, pH, and modified temperatures, all classified by the FDA (Food and Drug Administration) as safe and therefore useful in the food industry (Verma et al, 2021). For example, Lactobacillus plantarum synthesizes short-chain fatty acid esters with fewer than 10 carbon atoms; its optimal pH and temperature are 6.5 and 45°C, respectively. In the case of Malbranchea Cinnamomea, a high specificity for short- and medium-chain triglycerides has been reported; its optimal temperature and pH are 40 °C and 7.5, respectively. Streptomyces violascens is capable of tolerating organic solvents, exhibits enhanced regioselectivity at the sn-1 and sn-3 positions, and is highly specific, efficient, and selective during the hydrolysis of esters (Uppada et al. 2017; Duan et al, 2019; Gao et al, 2020) In this context, the analysis of triglycerides in vegetable oils using a biosensor could be beneficial for the fats and oils industry, as it would facilitate the control of hydrolysis as an indicator of their quality and stability. The detection mechanism is based on the reduction of glycerol in the presence of the enzyme lipase, forming triglycerides as a result of hydrolysis. The electrochemical reduction of oxygen present in the medium occurs at the Ag electrode. The potential difference during the enzymatic reaction (eq. 1) is recorded at the electrode and is proportional to the concentration of glyceryl tributyrate; this signal is amplified so that it can be processed (Fig. 1).
Fig. 1: Schematic representation of the key principle behind the biosensor's operation.
Lipase
Glycerol + 3 Fatty acids → Glyceryl tributyrate + H₂O (eq.1)
There are different methods, some of them very complex, for analyzing the products of hydrolysis in fats, as well as fatty acids. For example, a method was recently developed to quantify fatty acids in vegetable oils and non-dairy foods using capillary zone electrophoresis with UV detection. This technique combines CZE-UV with a column injection program and a single-point standard addition method (Leal de Oliveira et al, 2025). Short-chain fatty acids have also been analyzed using ultra-high-performance liquid chromatography-mass spectrometry (LC-MS/MS). The method allowed quantitative analysis of propionate, butyrate, isobutyrate, and valerate with acceptable selectivity, precision, and accuracy (Neef et al, 2026). Thus, the objective of this work is to develop a biosensor as a simple and valid method of analysis for monitoring in industrial plants during quality control of vegetable oils.
Materials and Methods
Chemicals and reagents LPS EC. 232-619-9 from Candida rugosa, with each vial containing 2.9 mg of protein, L-lactate, glutaraldehyde (50%), L-(+) lactic acid, HPLC grade sulfuric acid, glyceryl tributyrate, tricaprilin, tricaprine, trilaurin and flavin adenine dinucleotide were from Sigma (Sigma-Aldrich, St Louis, MO, USA), the preactivated immunodyne ABC membrane (Nylon 6, 6 pore size 0.45 µm) was supplied by Pall Europe (Porsmounth, UK). Sodium phosphate and anhydrous sodium acetate were obtained from Panreac (Panreac Química, Barcelona, Spain). Milli-Q deionized water was used throughout.
Measurement of Enzyme Activity and Preparation of the Candida Rugosa Lipase Solution The commercial enzyme was dissolved in 5 mL of 50mM buffer solution at pH 7, with an activity of 30 U/mg aliquoted in 0.5 mL Eppendorf tubes and frozen stored at -45ºC until use. This activity was analyzed with p-nitrophenyl palmitate at 37ºC at 200 rpm for 5 minutes. The supernatant was dissolved 10 times and read at an absorbance of 410 nm in a UV spectrophotometer (Dos Santos et al, 2025). The absolute activity was calculated using the following equation (eq. 2), while the specific activity was calculated using the following formula (eq.3) :
A = (ΔA / ε) · (V / t) · (1 / [E]) (eq. 2)
Where: A is the absolute lipase activity (in units of activity, such as U/mL). ΔA is the change in absorbance at a specific wavelength ԑ is the molar extinction coefficient of the reaction product V This is the reaction volume in mL t is the response time in minutes ⦋E⦌ is the enzyme concentration in mg/mL
Specific activity (U/g protein) = (total lipase activity) / (lipase protein content) (eq. 3)
Equipment The sensor used was Rank Brothers, Bottisman, Cambridge, England Model 20. The equipment used the enzyme immobilized on a nylon membrane. Its operation depends on two electrodes: the first contains a platinum disc with a diameter of 2 mm, and the second electrode is a reference electrode with a silver ring (Ag/AgCl). The sensor is connected to a terminal (computer) where the voltage change (mV) is recorded for further processing. YL9100 equipment manufactured by Young Lin Instrument CO., LTD (Korea) was used for chromatographic analysis.
Sample preparation It was necessary to make a water-oil emulsion so that during the hydrolysis of coconut oil a favorable microenvironment would form, which would increase the reaction speed and the final conversion of the oil hydrolysis. The optimal ratio was 1:2 (10mL water to 20 mL oil). The mixture was stirred (1, 000 rpm x 10 minutes) to form the appropriate emulsion. The reaction took place by adding 0.046 g of lipase to the well-mixed solution. The supernatant containing the hydrolyzed products was used for analysis.
Biosensor preparation and operation The electrode base allows the reactor to be adjusted at the top. This assembly allows the platinum electrode to receive the electron transfer and capture the signal, which is amplified in the transducer. To adjust the sensitivity of the instrument, the reaction cell is filled with distilled water saturated with oxygen and stirred until 100% saturation is reached. The entire process takes less than 10 seconds. The method was described in detail by Jadán et al, 2025; and Vergara et al, 2026.
Immobilized enzyme sensor The concentration of glutaraldehyde for the LPS enzyme was studied within a range of 5% to 15% and was set at 8%. Because this reagent forms a covalent bond between the membrane surface and the enzyme, it enables its immobilization. Thus, the total mixture (100 µL) contained 40 µL of LPS solution (30 U/mg), 50 µL of 0.1 M sodium phosphate buffer, pH 7.0, and 10 µL of glutaraldehyde (at a concentration of 8%). This mixture was then applied drop by drop onto a 1 cm² Immuno-Dyne ABC membrane and allowed to dry for 1 hour. After drying, 5 µL of 2% cellulose acetate solution was added to prevent interference with the matrix (Qiong et al., 1998).
Description of the chromatographic method For the analysis, a 40:60 acetone/acetonitrile mobile phase is used at a flow rate of 1.5 mL/min under isocratic conditions at 40°C. The column was a Lichrosorb RP 18, 5 µm x 4.6 mm x 25 cm. The equipment used is equipped with a quaternary pump, column oven, and automatic injector with a refractive index (RI) detector. Quantification was performed using a calibration curve between 1 mM and 5 mM glyceryl tributyrate. This is because, within this measurement range, the sensor's R² (coefficient of determination) allowed the values to be compared with those from HPLC by establishing a strong correlation between the measured variables.
Use of the enzyme sensor to analyze oil samples The enzyme sensor was used to quantify glyceryl tributyrate in previously hydrolyzed coconut oil. The method was validated and its results were compared with those obtained by high-performance liquid chromatography (HPLC).
Validation tests Reproducibility was established by injecting 1 mM glyceryl tributyrate standards or samples of the hydrolyzed solution into the immobilized enzyme system 30 consecutive times on the same day, using the same equipment and under the same conditions. The stability of the membrane stored at different temperatures was evaluated. Linearity was achieved by analyzing triplicate standards ranging from 1 to 5 mM for the immobilized enzyme system using the ordinary least squares (OLS) regression method. The sample dilution was 1/10 to obtain a strong signal, avoiding interference. To determine the stability of the membrane under operating conditions, successive injections of 5 mM glyceryl tributyrate were applied to a single membrane and the response was recorded until 50% of its initial activity was observed. The results were expressed as a function of time. The correlation between the sensor and HPLC was compared using different concentrations. Statistical methods. The increase in standard concentrations is analyzed using Statgraphics Plus software (v5.1). The linearity of the data between the sensor and the HPLC is determined by the coefficient of determination (R²) and the standard error (SE). A two-sample t-test (two-tailed) is used to determine if the HPLC and biosensor are statistically equivalent. The null hypothesis states that the population means are the same (H0: µ sensor = µ HPLC) while the alternative hypothesis states that the means are not equal (H0: H0: µ sensor ≠ µ HPLC).
Results
During the enzymatic reaction between LPS and the substrate glyceryl tributyrate, the oxygen present is reduced, resulting in electron transfer. This electrical signal is detected by the sensor electrode and is directly proportional to the concentration of glyceryl tributyrate in the coconut oil sample (eq.1). The signal originates in the silver ring where electron transfer is detected during oxygen reduction and is subsequently amplified for reading on the computer (Fig. 1). From this electron transfer (mV/s), the enzymatic reaction rate is determined using the slope (m) calculated at 7 seconds. The least interference in the response was observed at 7 seconds with an applied potential of -600 mV, minimizing background noise. The analysis of variability was determined by calculating the coefficients of variation (CV) in the hydrolysates and in the standards with consecutive injections, which were 3.75% (N=10) and 2.35% (N=9), respectively. The method was validated by evaluating repeatability, expressed as a coefficient of variation and analyzed in the sensor by simultaneous injections on the same day using identical conditions and a new membrane with immobilized enzyme. The results indicate values of 1.3% to 1.9% for repeatability (n=30), confirming acceptable accuracy and precision when quantifying glyceryl tributyrate in the oily hydrolysate. The possible interference of different triglycerides in the system was studied using reduction with the soluble enzyme at 37°C, followed by HPLC analysis. The results indicate that tricaprylin exhibited the highest relative oxidation percentage among the tested triglycerides after glyceryl tributyrate (Table 1). When tested with the biosensor, both trilaurin and tricaprylin showed a measurable response, suggesting a potential contribution to signal interference. However, these variations did not prevent normal sensor performance. The pH range studied was between 6 and 9 using 0.05 M phosphate buffer. Maximum enzymatic activity was observed at pH 7. The reaction temperature was set at 37°C, and these conditions were maintained throughout all experiments. No significant effect of pH on sensor sensitivity was observed under the tested conditions (data not shown).
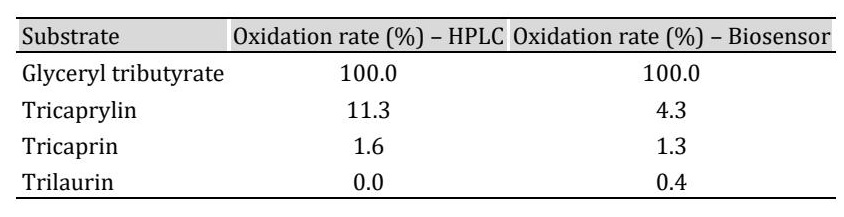
Table 1: Enzyme reduction rate in some triglycerides, measured with HPLC and the biosensor
Biosensor Features The kinetic parameter that determines enzyme affinity for a substrate is the Michaelis–Menten constant (Km). As shown in Fig. 2, increasing substrate concentration (glyceryl tributyrate) resulted in an increase in enzymatic reaction rate until a plateau was reached. At higher substrate concentrations (>2.5 mM), saturation of the enzyme active sites likely occurred. When the same analysis was performed with the immobilized enzyme, a reduction of approximately 15% in activity was observed, possibly due to incomplete coupling with the porous membrane surface. The calculated Km value was 0.13 mM, indicating high affinity of the enzyme–substrate complex. The Michaelis–Menten constant was calculated using the reference equation (eq.4), where Vmax values derived from Fig. 2 were used to determine this kinetic parameter.
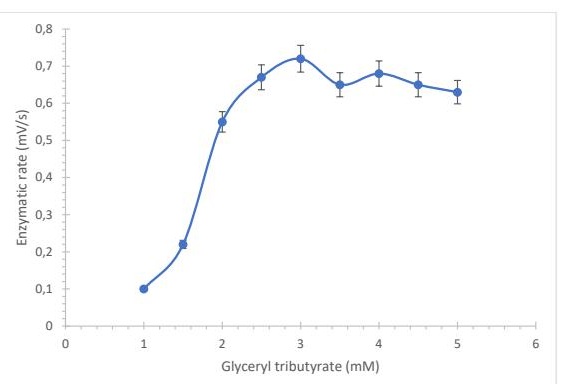
Fig. 2: Enzyme sensor response represented as the enzyme reaction rate as a function of glyceryl tributyrate concentration (n = 3).
S / v = (KM / Vmax) + (S / Vmax) (eq. 4)
LPS Mechanism for Reducing Glyceryl Tributyrate The affinity of LPS for the substrate was evaluated by reacting equal volumes (1, 500 µL) of enzyme solution (30 U/mg) with glyceryl tributyrate under agitation at 37°C. Aliquots were taken every 15 seconds until complete reduction of the substrate was observed. Total depletion occurred at 120 s, confirming high enzyme affinity (Fig. 3).
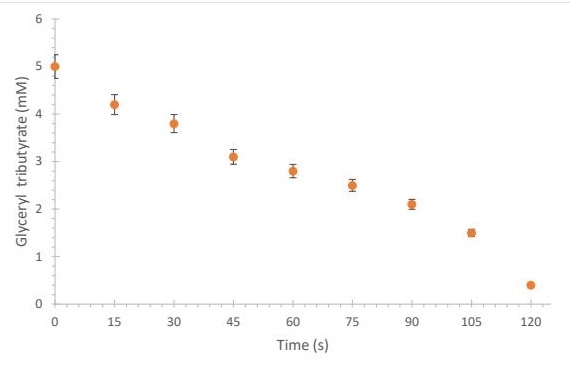
Fig. 3: Determination of glyceryl tributyrate in the presence of the enzyme (LPS) atpH 7.0 and 37°C. The points were determined by HPLC.
Calibration Curves The linear measurement range allowed determination of the detection limit (1 mM) and quantification limit (5 mM) using LPS as a biorecognition element. The correlation was strong, with a coefficient of determination R² = 0.998, indicating excellent agreement with HPLC measurements (Fig. 4). Repeatability was evaluated by 25 consecutive injections of glyceryl tributyrate standards (1 mM) and oil hydrolysates under identical conditions, using a fresh membrane for each measurement. The coefficients of variation ranged between 1.2% and 2.3%, confirming high precision and stability of the system.
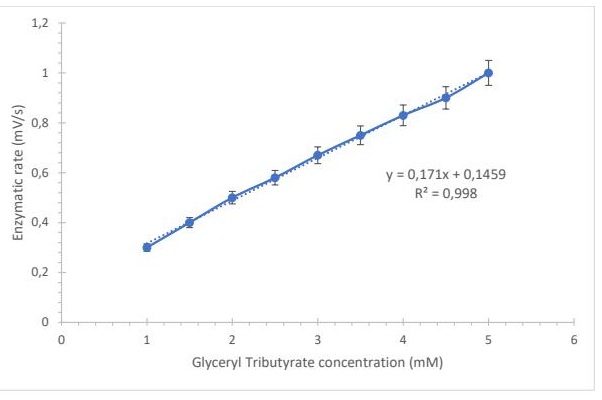
Fig. 4: Calibration curve obtained with an LPS biosensor with immobilized enzyme.
Glyceryl Tributyrate Measurements Using the Biosensor Compared to HPLC The biosensor was applied to determine glyceryl tributyrate concentration during oil hydrolysis. Four diluted samples (1:10) were analyzed and compared with HPLC measurements. A strong correlation was observed (R² = 0.9964) with a standard error of 0.015. A two-sample t-test (α = 0.05) showed no statistically significant difference between biosensor (Ẋ = 1.4, s = 0.1316) and HPLC measurements (Ẋ = 1.46, s = 0.1652), with t = 0.0451 (df = 30) and p = 0.0765. This agreement was further confirmed across 20 samples (Table 2).
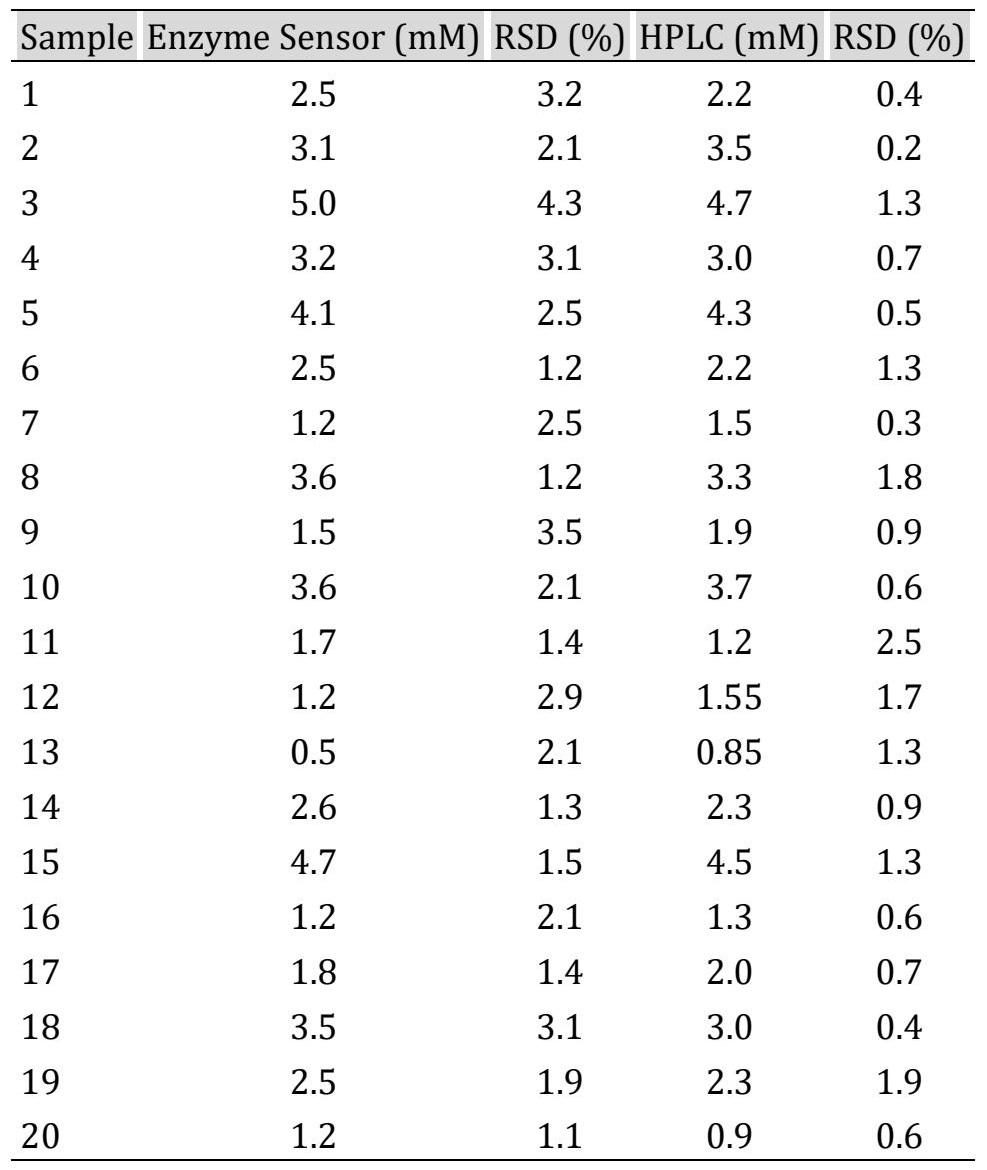
Table 2: Measurements of glyceryl tributyrate levels by the enzyme sensor and High Performance Liquid Chromatography. Results are expressed in mM ± CV (n=3)
Stability of the Immobilized Enzyme The stability of the enzyme immobilized on a nylon membrane was evaluated over 280 days at 4°C. Measurements were performed at 5-day intervals. The enzyme retained approximately 90% of its initial activity for up to 260 days, after which a decline was observed, likely due to conformational changes and alterations in the active site. The membrane could be reused up to 27 times without significant loss of activity, demonstrating good long-term stability (Fig. 5).
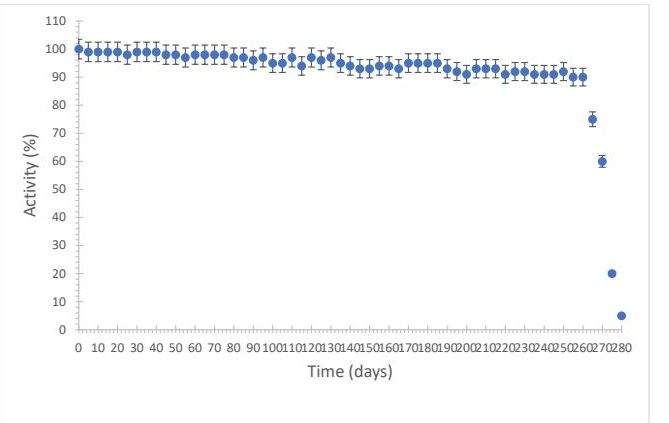
Fig. 5: Stability of lipase in solution stored under refrigeration (4 °C). n = 3.
Discussion
The developed biosensor demonstrates reliable performance for the quantification of glyceryl tributyrate in vegetable oils. The observed low variability and high repeatability indicate a robust analytical system with minimal matrix interference. The strong agreement with HPLC results further supports its analytical validity. The interference study showed that, although tricaprylin exhibited the highest relative oxidation among the tested triglycerides, overall interference remained limited and did not significantly affect sensor performance. This suggests a higher affinity of the enzyme toward glyceryl tributyrate compared to other triglycerides, enabling near-selective detection in complex matrices. Compared to conventional analytical techniques such as near-infrared spectroscopy, electrochemical methods using indium tin oxide electrodes, and liquid chromatography, the proposed biosensor offers advantages including reduced reagent consumption, faster response time, and lower environmental impact. Reported detection ranges for these techniques are comparable (16.07 mg dl⁻¹; 0.56–3.39 mM; 10–80 mg/L, respectively) (Oliveira Neves et al., 2012; Bhardwaj et al., 2015; Carballo et al., 2019). Although the detection limit of the biosensor is relatively high compared to clinical analytical methods, it is suitable for industrial quality control applications, where triglyceride concentrations are typically significantly higher. This limitation does not affect its practical applicability in routine monitoring of vegetable oils. The proposed system therefore represents a practical and cost-effective alternative for routine industrial monitoring.
Conclusion
An analytical method was developed for the quantification of glyceryl tributyrate in vegetable oil hydrolysates. The method showed enzymatic stability for 260 days, during which time the loss was not significant, so it was able to recognize the analyte of interest. During operation, the sensor with the enzyme (LPS) showed high selectivity towards the substrate KM= 0.13 mM. The sensor results were comparable to those obtained by high-performance liquid chromatography (HPLC), with the exception of costs and times (lower for the sensor). In conclusion, the sensor was reliable R2= 0.998, after optimizing all variables and values associated with detection and quantification.
Acknowledgements
Financing
This work has not received any funding.
Data availability
Data will be made available upon request.
Disclosure Statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Bhardwaj, S.K; Mahapatro,, A.K; Basu, T. 2015. Bienzymatic triglyceride biosensor based on electrochemically reduced graphene oxide. Int. J. Chem Tech Res. 7, 858-866.
- Carballo, S; Prats, M; Maestre, S. 2019. Microwave assisted high performance liquid chromatography for the separation of triacylglycerols in vegetable oils using an evaporative light scattering detector. Food Chemistry. 300, 125203 https://doi.org/10.1016/j.foodchem.2019.125203
- Dos Santos, L.A; Krieger, N; Mitchell, D.A; Mateo, C. 2025. Triton X-100 modified polygalacturonic acid: A novel coating for enhanced activity of immobilized lipases. Carbohydrate Polymers. 370, 124140. https://doi.org/10.1016/j.carbpol.2025.124140
- Gao, K; Chu, W; Sun, J; Mao, X. 2020. Identification of an alkaline lipase capable of better enrichment of EPA than DHA due to fatty acids selectivity and regioselectivity. Food Chemistry. 330 (5).127225. https://doi.org/10.1016/j.foodchem.2020.127225
- Jadán, F; Villavicencio, Ch; Mieles, J; Cantos, M; Anchundia, A; Zambrano, M; Intriago, F; Giler, S; Pérez, E; Litardo, R; Jadán, C. 2025. Quantification of lactic acid in wines using an amperometric biosensor. Food Control. 167, 110821. https://doi.org/10.1016/j.foodcont.2024.110821
- Leal de Oliveira, M; Queiroz de Souza, J; Nascimento, M; Oliveira Moreira, O; Geraldo, P. 2025. Determination of trans - fatty acids in non-dairy foods using enhanced CZE-UV: Implications for food health assessment. Journal of Food Composition and Analysis. Volume 148, Part 2, 108341 https://doi.org/10.1016/j.jfca.2025.108341
- Liu, Y; Yamaguchi, Sh; Ishigaki, Y; Chen, J; Liu, X; Li, J; Zhou, Z. 2025. Protease-lipase co-mediated cheese microstructural changes enhanced proteolysis and lipolysis kinetics during in vitro dynamic digestion for the elderly. Food Chemistry. 493, 146045 https://doi.org/10.1016/j.foodchem.2025.146045
- Liu, J; Zheng, Y; Zhang, R; Yu, Y; Wang, F; Deng, L; Wu, K. 2024. A novel synthesis method of medium and long chain triglyceride lipids from rubber seed oil catalyzed by enzymatic interesterification and its metabolism mechanism. Food & function. 15 (19), 9903-9915. https://doi.org/10.1039/D3FO05662G
- Narwal, V; Pundir, C.S. 2017. An improved amperometric triglyceride biosensor based on co-immobilization of nanoparticles of lipase, glycerol kinase and glycerol 3-phosphate oxidase onto pencil graphite electrode. Enzyme and Microbial Technology. 100, 11-16. https://doi.org/10.1016/j.enzmictec.2017.01.009
- Neef, S; Opara, J; Heinkele, G; Gross, M; Hofmann, U; Schwab, M; Zimmermann, M; Haag, M; Mürdter,T. 2026. Quantitative analysis of underivatised short-chain fatty acids and simultaneous non-targeted metabolic profiling by ultra-high performance liquid chromatography-mass spectrometry. Microchemical Journal. Volume 221, 116757. https://doi.org/10.1016/j.microc.2025.116757
- Oliveira Neves, A; Atanues de Araujo, A; Laís, B; Valderrama, P; Marco, P; Gomes de Lima, K. 2012. Near infrared spectroscopy and multivariate calibration for simultaneous determination of glucose, triglycerides and high-density lipoprotein in animal plasma. Journal of Pharmaceutical and Biomedical Analysis. Volume 66, 252-257. https://doi.org/10.1016/j.jpba.2012.03.023
- Qiong, C; Tuzhi, P; & Liju, Y. 1998. Silk fibroin/cellulose acetate membrane electrodes incorporating xanthine oxidase for the determination of fish freshness. Analytica Chimica Acta. 369, 245-251. https://doi.org/10.1016/S0003-2670(98)00213-X
- Shi, J; Zhu, J; Yang, Y; Qiao, S; Dai, H; Chen, H; Ma, L; Zhang, Y; Wang, H. 2025. Lipase etching effects-induced interaction between betanin nanocomplexes and polylactic acid and its 3D printing for food packaging. Food Chemistry. 464, 141636. https://doi.org/10.1016/j.foodchem.2024.141636
- Uppada, S.R; Akula, M; Bhattacharya, A; Dutta, J.R. 2017. Immobilized lipase from Lactobacillus plantarum in meat degradation and synthesis of flavor esters. Journal of Genetic Engineering and Biotechnology. 15(2), 331-334. https://doi.org/10.1016/j.jgeb.2017.07.008
- Vergara, G; Real, G; Santana, G; Caballero, H; Litardo, R; Noriega, D; Anchundia, A; Arcentales, Ch; Peña, A; Vera, J; Jadán C; Jadán, F. 2026. Selective quantification of tryptophan in milk using a sensor based on L-tryptophan dehydrogenase. Food Science and Biotechnology.
- Verma, S; Meghwanshi, G.K; Kumar, R. 2021. Current perspectives for microbial lipases from extremophiles and metagenomics. Biochimie.182, 23-36. https://doi.org/10.1016/j.biochi.2020.12.027
- Xiang, X; Wang, K; Wang, F; Yang, Q; Huang, J; Zhou, Q; Wang, Q. 2025. Enhancing beef tallow flavor through enzymatic hydrolysis: Unveiling key aroma precursors and volatile compounds using machine learning. Food Chemistry. 477. 143559. https://doi.org/10.1016/j.foodchem.2025.143559
- Zhang, M; Xu, B; Zhao, D; Shen, M; Li, M; Liu, D; Liu, L. 2024. Production of margarine fat containing medium and long chain triacylglycerols by enzymatic interesterification of peony seed oil, palm stearin and coconut oil blends. Foods. 13(9),1405. https://doi.org/10.3390/foods13091405
- Zhong, H; Jin, J; Zhou, Q; Zhang, Y; Zheng, M. 2024. Construction of a Pickering interfacial biocatalysis system in skim milk and enzymatic transesterification for enhancement of flavor and quality. Journal of Dairy Science. 107(11). 9074-9086. https://doi.org/10.3168/jds.2024-25037
- Zou, X; Khan, I; Wang, Y; Hussain, M; Jiang, B; Zheng, L; Pan, Y; Hu, J. 2024. Preparation of medium and long chain triacylglycerols rich in n-3 polyunsaturated fatty acids by bio-imprinted lipase-catalyzed interesterification. Food Chemistry. 455, 139907. https://doi.org/10.1016/j.foodchem.2024.139907