Original Article – DOI: 10.33594/000000862
CPB (60): 199 - 212
Accepted: 15.04.2026 - Published: 24.04.2026
Ultra-Broadband Micromechanical Ultrasound (UMUS) as a Strategy to Correct Cyclophosphamide-Induced Myelosuppression Without Limiting Antitumor Efficacy
bEvolus Technologies, Haifa 3551334, Israel
Keywords
Abstract
Background/Aims: This study aimed to apply ultra-broadband micromechanical ultrasound (UMUS) for correction of myelosuppression caused by the cytotoxic effects of cyclophosphamide without limiting its antitumor efficacy. Methods: The study included animals bearing transplanted Ehrlich carcinoma. Cyclophosphamide (CP) was administered once daily for three consecutive days starting on day 8 of tumor growth at a cumulative dose of 330 mg/kg per mouse. After completion of CP administration, a subset of animals was exposed to UMUS irradiation once daily for five days. Control groups included mice without tumors and tumor-bearing mice not exposed to CP or UMUS. Tumor growth kinetics were analyzed, and quantitative parameters of peripheral blood, bone marrow, and spleen were determined. Results: The obtained data indicate that UMUS exposure does not reduce the antitumor efficacy of CP but is associated with enhanced recovery of the hematopoietic system the antitumor efficacy of CP but exerts a positive effect on the recovery of the hematopoietic system in the bone marrow. This is confirmed by a statistically significant increase in the number of cells (compared with animals not exposed to UMUS) in specific bone marrow hematopoietic pools, including myelokaryocytes, blast cells, erythroid, lymphoid, and megakaryocytic cells. Conclusion: UMUS exposure was associated with accelerated recovery of multiple hematopoietic lineages in the bone marrow following cyclophosphamide-induced injury, without compromising antitumor efficacy. These findings suggest that UMUS may represent a promising non-pharmacological strategy to support hematopoietic regeneration during cytotoxic chemotherapy.
Introduction
Anticancer chemotherapy is known to be characterized by relatively low specificity of antitumor action and high toxicity toward normal organs and tissues. This is primarily due to damage to rapidly proliferating cells, particularly the epithelium of the gastrointestinal tract and hematopoietic tissue [1-3]. Most cytostatic agents are cell cycle–specific or phase-specific and exhibit maximal cytotoxic activity against actively proliferating cells. This property underlies their therapeutic index but simultaneously determines their toxicity profile [4-5]. Bone marrow damage and the development of hemato- and myelotoxicity (leukopenia, anemia, and thrombocytopenia) are among the most common and clinically significant complications of anticancer chemotherapy [6, 7]. Beyond the direct threat to patients [8, 9] damage to the hematopoietic system represents one of the key factors limiting the effectiveness of antitumor therapy. Therefore, correction of hematological toxicity and the search for new methods to protect bone marrow remain important challenges in modern oncology. In clinical practice, granulocyte colony-stimulating factors (G-CSF), such as filgrastim and pegfilgrastim, are widely used for the prevention and treatment of neutropenia in accordance with the recommendations of the National Comprehensive Cancer Network [10]. However, the use of G-CSF may be associated with adverse effects (bone pain, splenomegaly, and rare cases of capillary leak syndrome), as well as significant economic costs for healthcare systems [11]. Iron preparations and erythropoiesis-stimulating agents (ESA) are used to correct anemia; however, their use is limited due to the risk of thromboembolic complications and potential effects on oncological outcomes, as reflected in the recommendations of the American Society of Clinical Oncology and the American Society of Hematology [12, 13]. Pharmacological correction of thrombocytopenia remains challenging: thrombopoietin receptor agonists are being investigated, but their routine use in chemotherapy-induced thrombocytopenia is limited by insufficient evidence and potential risks [14]. Thus, the effectiveness of pharmacological supportive agents is relative and not devoid of risks. Their use is often accompanied by adverse effects, requires careful monitoring, and is associated with significant economic costs [15, 16], highlighting the need to develop new approaches for the prevention and correction of myelotoxicity without compromising the antitumor efficacy of treatment. In addition to pharmacological approaches, increasing attention has been directed toward physical and mechanobiological strategies capable of modulating stem cell function and tissue regeneration. Low-intensity ultrasound has been shown to influence cellular proliferation, differentiation, and microenvironmental signaling [17, 18]. However, its potential to selectively enhance hematopoietic recovery following chemotherapy-induced myelosuppression without interfering with antitumor efficacy remains insufficiently explored. One promising approach is the use of ultra-broadband micromechanical ultrasound (UMUS), which enables modulation of the biological effects of ultrasound exposure through an expanded frequency spectrum at minimal energy load [19]. It should be noted that regardless of the nature of supportive therapy — whether pharmacological agents, physicochemical, or physical corrective methods — such interventions must meet several key requirements: (i) they must not stimulate tumor progression; (ii) they must not reduce the effectiveness of antitumor therapy; (iii) they must correct the manifestations of treatment-related toxicity. Therefore, the aim of this study was to investigate the effect of ultra-broadband micromechanical ultrasound (UMUS) on the efficacy of antitumor therapy and its ability to correct manifestations of myelosuppression induced by cytotoxic anticancer drugs. Cyclophosphamide, an alkylating cytotoxic agent characterized by a broad spectrum of toxicity toward normal organs and tissues, among which hematological toxicity is dose-limiting, was used.
Materials and Methods
Study Design In vivo studies were conducted using female C57Bl/6 mice (80 animals in total) aged 10–12 weeks and weighing 22–25 g, bred in the vivarium of IEPOR NASU. All procedures involving animals were conducted in accordance with institutional guidelines for the care and use of laboratory animals and were approved by the Bioethics Committee of IEPOR NASU (protocol No. 4, dated 04/03/2025). Animals were housed in groups under standard controlled conditions with a 12-h light/dark cycle and were provided with standard rodent chow and water ad libitum. Throughout the experiment, mice were weighed three times per week and their general condition was monitored. The study included three groups of animals bearing transplanted Ehrlich carcinoma (Fig. 1a). Tumor cells were expanded in vitro and inoculated subcutaneously into the dorsal interscapular region at a dose of 1.5 × 10⁶ cells per mouse. After tumor inoculation, animals were randomized by body weight and divided into three groups: negative control, positive control, and experimental. Animals in the positive control and experimental groups received intraperitoneal cyclophosphamide (CP) at a dose of 110 mg/kg/day in 0.5 ml saline once daily for three consecutive days beginning on day 8 of tumor growth. The cumulative dose was 330 mg/kg per mouse (corresponding to the maximum tolerated dose, MTD). Mice in the negative control group received 0.5 ml of saline intraperitoneally at the same time points. Animals in the experimental group were exposed to UMUS once daily for five days beginning on day 11 after tumor transplantation. The target area for ultrasound exposure was the hind limb of the animals (region of the femur and tibia). Prior to ultrasound treatment, hair on the limbs of mice in all groups was removed by depilation, and ultrasound exposure was initiated 24 h after depilation. Ultrasound gel (Aqua Ultra Basic UBQ 5000 Ultragel, Hungary) served as the coupling medium between the animal tissue and the ultrasound emitter. On days 11, 16, and 23 of tumor growth, animals were sacrificed by decapitation under ether anesthesia, and quantitative parameters of peripheral blood, bone marrow, and spleen were analyzed. Hematological parameters were determined in five intact mice and in five mice of each tumor-bearing mouse group at days 11, 16, and 23 of tumor growth.
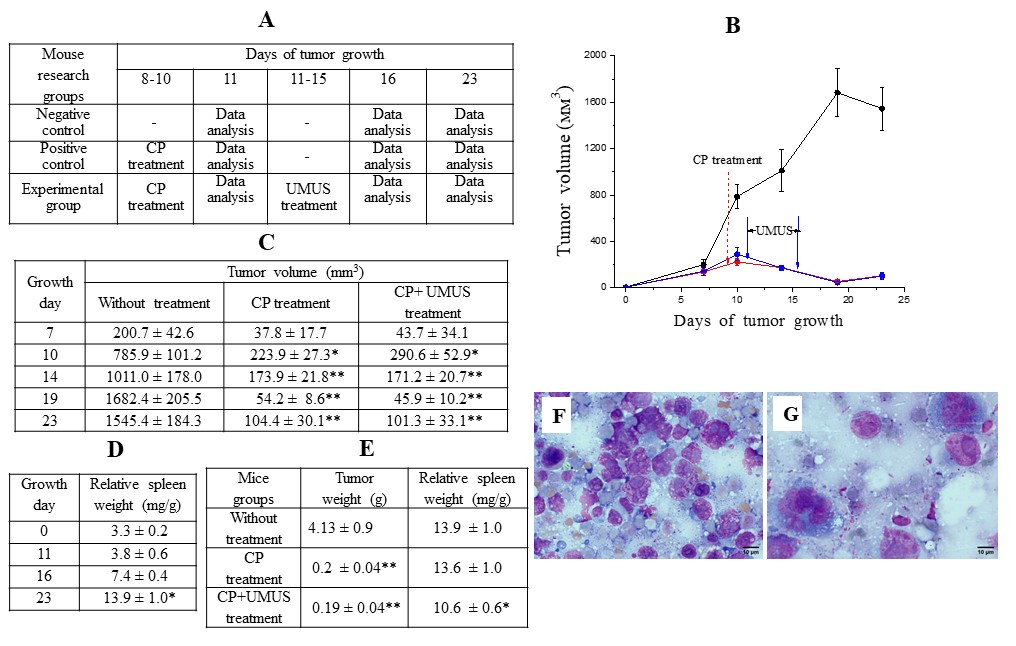
Fig. 1: Effect of UMUS on the antitumor efficacy of cyclophosphamide (A) Study design; (B) and (C) Tumor growth kinetics in animals without exposure to CP and UMUS (black symbols) and after exposure to either CP (red) or CP + UMUS (blue); (D) Dynamics of relative spleen weight during tumor growth; (E) Tumor weight and relative spleen weight on day 23 of growth after treatment with either CP or CP + UMUS; (F) Tumor imprint smears on day 11 of growth without CP treatment; (G) Tumor imprint smears on day 11 of growth after CP treatment. * (p<0.05) and ** (p<0.01)– statistically significant differences compared with the corresponding values in the absence of CP and UMUS.
Ultrasound Parameters A signal generator of a special waveform (SDG 2082X Siglent generator, USA) was used as the ultrasound generator. Frequency range: 1–15 MHz. Intensity: 0.22 mW/cm². Pulse repetition rate: 1 kHz. Exposure time: 5 min.
Evaluation of Ehrlich Carcinoma Growth Kinetics Tumor size was measured in three mutually perpendicular planes on days 7, 10, 14, 19, and 23 of tumor growth. Tumor volume (V, mm³) was calculated according to the formula:
V = L × W × D × π/6 (1)
where L is length, W — width, D — thickness, π = 3.14.
Peripheral Blood Parameters Peripheral blood samples were collected after decapitation into tubes containing K3-EDTA (Amed, China) and thoroughly mixed. Hematological parameters were measured using a BC-5000Vet hematology analyzer (Mindray, China) according to the manufacturer’s instructions.
Bone Marrow Hematopoiesis Analysis Bone marrow for quantitative analysis was flushed from the femur with 1 ml of physiological saline. The number of myelokaryocytes was determined using the standard counting method in a Goryaev chamber. Smears were prepared from bone marrow isolated from the tibia. The cellular composition of the myelogram was determined by counting 500 cells per preparation. Cells were analyzed in pooled hematopoietic compartments according to the differentiation grade and maturation: At the indicated time points, the spleen was removed and weighed. The relative spleen weight (M) was calculated according to the formula:
M = Ms/(Mm − Mt) (2)
where Ms is spleen weight; Mm — body weight of the mouse; Mt — tumor weight.
For cytological examination, the spleen was sectioned longitudinally and imprint smears were prepared. The cellular composition of the splenogram was determined by counting 300 cells per preparation. Smears of hematopoietic organs and tumor tissue were stained using the Pappenheim method and analyzed using a light immersion microscope at ×1000 magnification. Microphotographs were obtained using an Axio Imager A2 microscope (Carl Zeiss, Germany) equipped with an Axiocam 712 color digital camera.
Statistical Analysis Statistical analysis was performed using descriptive statistics as well as parametric (Student’s t-test for tumor growth kinetics analysis) and non-parametric (Mann–Whitney for statistical analysis of peripheral blood, bone marrow, and spleen parameters) tests using the software packages ORIGIN 9.5 and STATISTICA. Differences were considered statistically significant at p < 0.05. The data are presented as M±SE.
Results
Changes in Hematopoietic System Parameters during the Growth of Ehrlich Carcinoma Following inoculation of tumor cells, tumor formation and growth were observed in 100% of mice. Tumor progression during the study period did not result in animal mortality. Analysis of the growth kinetics of Ehrlich carcinoma (in the absence of CP and UMUS exposure) demonstrated progressive tumor growth until day 19, followed by stabilization thereafter (Fig. 1B, Fig. 1C). Against the background of progressive tumor growth, an increase in the relative spleen weight was observed beginning from day 11. By day 16, the relative spleen weight was nearly twofold higher than that of intact mice (p < 0.01), and by day 23 it was approximately threefold higher (p < 0.001) (Fig. 1D). Tumor growth was also accompanied by significant alterations in peripheral blood parameters, particularly in hemoglobin level (Fig. 2A), erythrocyte count (Fig. 2B), platelet count (Fig. 2C), and leukocyte count (Fig. 2D). Hemoglobin levels and erythrocyte counts significantly decreased by 11% and 15% on days 16 and 23 of tumor growth, respectively. These changes occurred against a background of morphological alterations characterized by increased mean erythrocyte volume and hemoglobin content per erythrocyte. Hematocrit and hemoglobin concentration in erythrocyte mass were slightly decreased compared with intact controls, which was mainly due to the reduction in erythrocyte numbers. In contrast to the erythroid parameters, platelet levels decreased significantly as early as day 11 of tumor growth. Platelet counts at this time point were 33% lower than in intact mice (p < 0.01). Despite temporary compensatory normalization observed on day 16, platelet levels at later stages of tumor growth remained 23% lower than normal (p < 0.05). The observed reduction in platelet levels is likely associated with the invasive nature of Ehrlich carcinoma growth. The most pronounced changes were observed in leukocyte levels in peripheral blood. By day 11, leukocyte counts decreased by 53% (p < 0.01). Subsequently, leukocyte counts increased, and by day 23 of tumor growth, they significantly exceeded the levels observed in intact controls (+57%). Tumor growth was accompanied by compensatory activation of hematopoiesis in the spleen, resulting in a significant increase in both granulocytic and erythroid hematopoietic pools (Fig. 2E, F). This progressive activation largely explains the marked increase in relative spleen mass observed on days 16 and 23 of tumor growth (Fig. 1D), emphasizing the important role of the spleen in hematopoietic recovery. Significant changes during tumor progression were also observed in the number of myelokaryocytes. Their number decreased more than twofold (p < 0.01) compared with intact animals beginning from day 16 of tumor growth (Fig. 3A-C). It should be noted that during the progression of Ehrlich carcinoma, isolated tumor cells were detected both in the bone marrow (Fig. 3D) and in the spleen (Fig. 3E), which correlates with the highly invasive nature of this tumor.
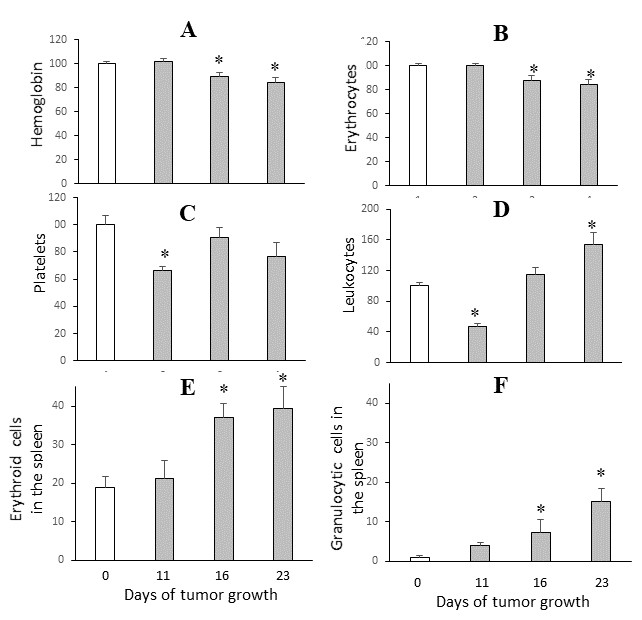
Fig. 2: Dynamics of changes in peripheral blood parameters in mice (as percentages of intact animal values) (A–D) and cellular composition of the spleen (E–F) during Ehrlich carcinoma growth. * (p<0.05)– statistically significant differences compared with intact mice.
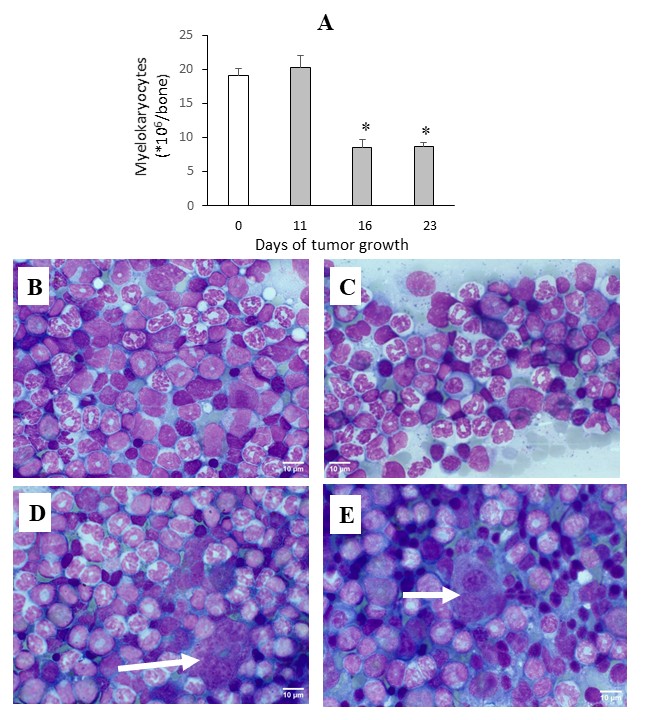
Fig. 3: Dynamics of changes in the number of myelokaryocytes during Ehrlich carcinoma growth (A). Bone marrow of intact mice (B) and mice with Ehrlich carcinoma on day 23 of tumor growth (C). Presence of Ehrlich carcinoma cells in bone marrow (D) and spleen (E). * (p<0.05) – statistically significant differences compared with intact mice.
Effect of UMUS on Ehrlich Carcinoma Growth and Peripheral Blood Parameters after Cyclophosphamide Therapy Administration of CP for three consecutive days at a cumulative dose of 330 mg/kg (corresponding to the MTD) beginning on day 8 of tumor growth resulted in marked inhibition of Ehrlich carcinoma growth (Fig. 1B, Fig. 1C). The tumor growth inhibition rate reached 71% on day 10 (p < 0.01) and progressively increased to 97% by day 19 of tumor growth (p < 0.0001). By day 23, the inhibition rate remained high at approximately 95%, both in terms of tumor volume and tumor weight (Fig. 1C, E). Importantly, the pronounced antitumor effect of CP was not accompanied by mortality of experimental animals. Notably, despite pronounced tumor growth inhibition, CP treatment did not significantly affect the relative spleen weight, which remained relatively high (Fig. 1E). The strong antitumor activity of CP was accompanied by significant changes in peripheral blood parameters (Fig. 4). A relatively moderate but statistically significant decrease in hemoglobin levels (approximately 10%) was observed already on day 11 (immediately after completion of CP administration) and persisted until day 23 of tumor growth (Fig. 4A). CP administration did not directly affect platelet counts, the reduced levels of which were primarily associated with tumor growth itself (Fig. 4B, Fig. 1C). The normalization of platelet levels observed on day 23 was most likely due to a substantial reduction in the tumor-induced negative impact on this parameter as a result of significant inhibition of tumor growth by CP. The most pronounced effect of CP on peripheral blood parameters concerned leukocyte levels (Fig. 4C). Immediately after completion of CP administration (day 11 of tumor growth), leukocyte counts did not exceed 12% of those observed in intact animals and were fourfold lower than in tumor-bearing mice not treated with CP. This reduction affected both neutrophils and lymphocytes; however, the decrease in lymphocytes was substantially greater (Fig. 4D). Five days after CP administration, a rapid and pronounced recovery of the leukocyte lineage was observed. By day 16, leukocyte counts exceeded those of intact mice by 60% (p < 0.01), indicating leukocytosis. By day 23, this parameter returned to the level observed in the control group. Analysis of the effect of UMUS on peripheral blood parameters in tumor-bearing animals after CP administration revealed no significant differences compared with animals receiving CP alone. By day 23 of tumor growth (two weeks after completion of CP treatment), complete normalization of hemoglobin and leukocyte levels was observed regardless of UMUS exposure, indicating a high regenerative capacity of bone marrow hematopoiesis. Notably, on day 16 of tumor growth, UMUS contributed to normalization of leukocyte levels, whereas recovery without UMUS resulted in pronounced leukocytosis. Recovery of peripheral blood cell pools is primarily determined by the functional capacity of the main hematopoietic organs — bone marrow and spleen.
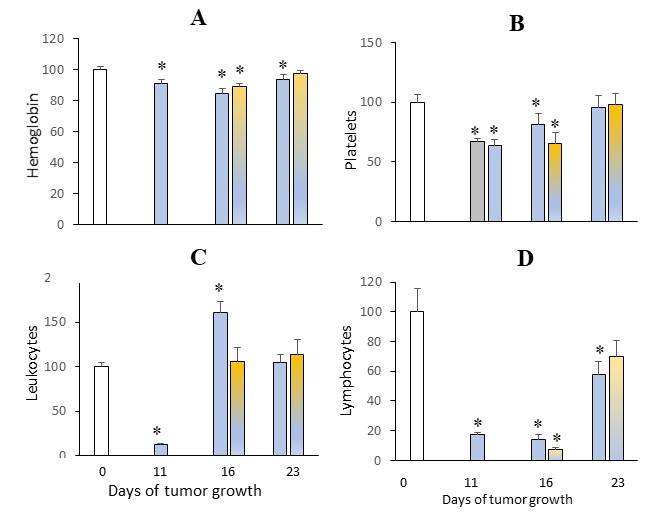
Fig. 4: Dynamics of changes in hemoglobin levels (A), numbers of platelets (B), leukocytes (C), and lymphocytes (D) (% of intact mice values – white bars) after CP administration (blue bars) without UMUS and with UMUS (yellow-blue bars). * (p<0.05) – statistically significant differences compared with intact mice.
Effect of UMUS on Bone Marrow Hematopoiesis Recovery after Cyclophosphamide Therapy The study demonstrated that CP administration exerted a pronounced effect on the bone marrow. After three consecutive CP administrations (day 11), bone marrow smears contained numerous signs of cellular destruction, including naked nuclei, cytolysis, karyolysis, pathological mitoses, and giant cells. The number of myelokaryocytes after completion of CP treatment (day 11) did not exceed 3% of normal values (Fig. 5A, C). Five days later, regenerative processes resulted in an increase in myelokaryocyte numbers; however, even on days 16 and 23 their levels did not exceed 50–55% of those observed in intact animals. Despite the low level of myelokaryocytes, two weeks after CP administration (day 23 of tumor growth), UMUS exposure resulted in a statistically significant increase of their number by 35–40% compared with animals not exposed to UMUS, reaching levels comparable to intact animals (Fig. 5E). This finding indicates that UMUS positively affects recovery of bone marrow hematopoiesis. Analysis of the effects of CP and UMUS on individual bone marrow hematopoietic pools further supports this conclusion. As shown in Fig. 6, UMUS exposure resulted in a statistically significant increase in blast cell numbers on both day 16 and day 23 of tumor growth compared to the corresponding indicator after CP treatment alone, which resulted in an approximately tenfold decrease in the absolute number of blast cells (Fig. 6A). By day 23, blast cell numbers reached levels comparable with intact controls, indicating a substantial increase in the regenerative potential of bone marrow hematopoiesis under the influence of UMUS. Analysis also revealed rapid restoration of the granulocytic pool, and by day 23 of tumor growth, granulocyte numbers did not differ significantly from those in intact animals (Fig. 6C). Recovery of the erythroid pool after its pronounced inhibition by CP on day 11 occurred more slowly. By day 16, erythroid cells constituted only about 7% of control levels, while their absolute number remained approximately tenfold lower than in intact animals (Fig. 6D). Acceleration of erythroid regeneration was observed only by day 23 of tumor growth, although erythroid cell numbers remained threefold lower than in intact animals. UMUS significantly accelerated recovery of the erythroid hematopoietic pool in the bone marrow, resulting in an almost twofold increase in erythroid cell numbers compared with animals receiving CP alone, reaching levels comparable with intact controls (Fig. 6D). Cyclophosphamide administration also resulted in an almost tenfold decrease in lymphoid cell numbers (Fig. 6E). UMUS stimulated recovery of the lymphoid lineage, so that by day 23 lymphoid cell numbers were nearly threefold higher than in animals receiving CP without UMUS, reaching levels comparable with intact controls. Pronounced bone marrow hypoplasia after CP administration also manifested as significant inhibition of the megakaryocytic pool. As shown in Fig. 6F, megakaryocyte numbers on day 11 were 37 times lower than in intact controls. UMUS induced a progressive increase in megakaryocyte numbers. By day 23, their number was approximately twofold higher than in animals receiving CP without UMUS; however, it still remained relatively low compared with intact controls (Fig. 6G, H).
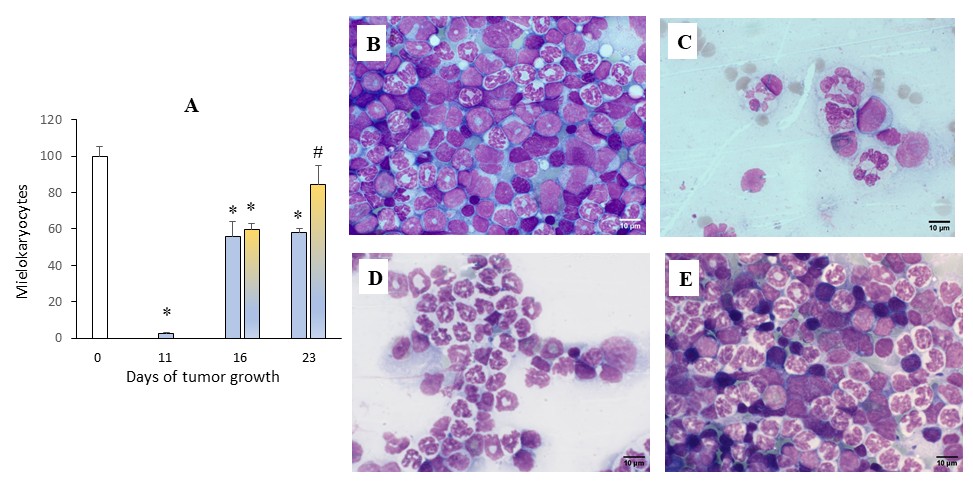
Fig. 5: Effect of CP and UMUS on the number of myelocytes during Ehrlich carcinoma growth. Dynamics of changes in the number of myelokaryocytes (% of intact mice values – white bars) after CP administration (blue bars) without UMUS and after UMUS (yellow-blue bars) (A). Bone marrow depletion after CP administration on day 11 of tumor growth (C) compared with myelokaryocyte content in tumor-bearing animals without CP exposure (B). Recovery of hematopoiesis in mice on day 23 of tumor growth after CP administration without UMUS (D) and after UMUS exposure (E). * (p<0.05) – statistically significant differences compared with intact mice. # (p<0.05) – statistically significant differences compared with corresponding value after CP treatment alone.
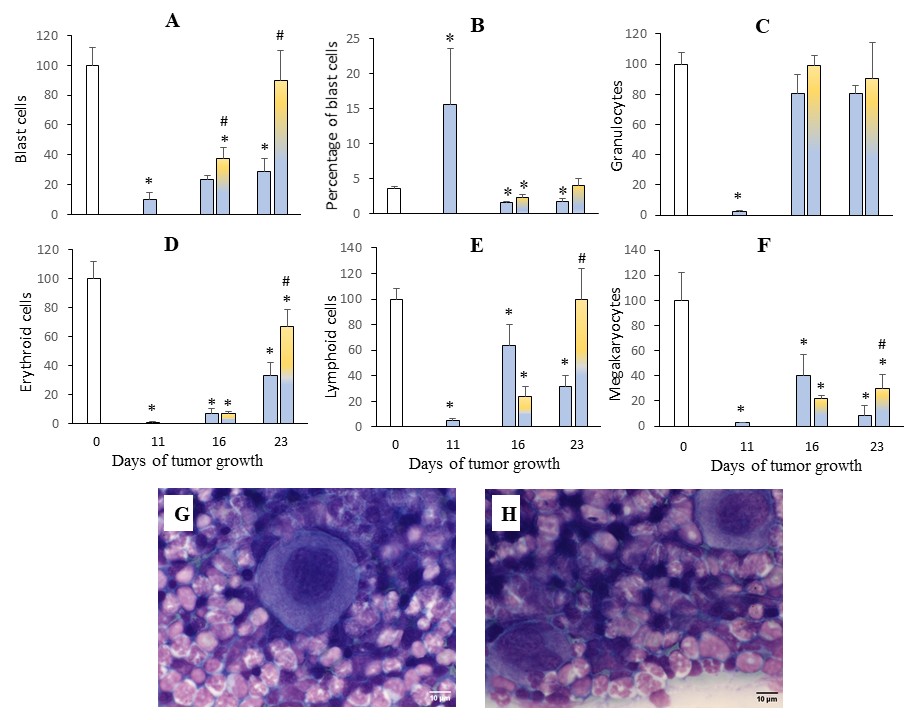
Fig. 6: Effect of UMUS on bone marrow hematopoiesis during CP therapy. Changes in the distribution of bone marrow cell pools according to the degree of differentiation and maturation (% of intact mice values – white bars) after CP administration (blue bars) without UMUS and after UMUS (yellow-blue bars) (A–F). Increase in the number of megakaryocytes (white arrows) after UMUS exposure on day 23 (H) compared with the corresponding value in animals after CP administration without UMUS (G). * – statistically significant differences compared with intact mice. # (p<0.05) – statistically significant differences compared with corresponding value after CP treatment alone.
Effect of UMUS on Recovery of Spleen-associated Hematopoiesis after Cyclophosphamide Therapy Tumor growth itself influenced hematopoietic processes in the spleen, increasing the proportion of granulocytic cells (from 1% to 15% by day 23) and erythroid cells (from 19% to 30%) (Fig. 7C). After CP administration (day 11 of tumor growth), destructive changes were observed in the spleen (Fig. 7B); however, the relative content of erythroid and granulocytic cells did not differ significantly from that observed in untreated tumor-bearing animals. Subsequently, in animals of both groups (with and without UMUS), a significant increase in the content of cells from the main hematopoietic lineages was observed compared with controls. No statistically significant differences were detected between groups with or without UMUS exposure. However, it should be noted that on day 23 animals exposed to UMUS showed a tendency toward reduced proportions of granulocytic and erythroid elements in the spleen.
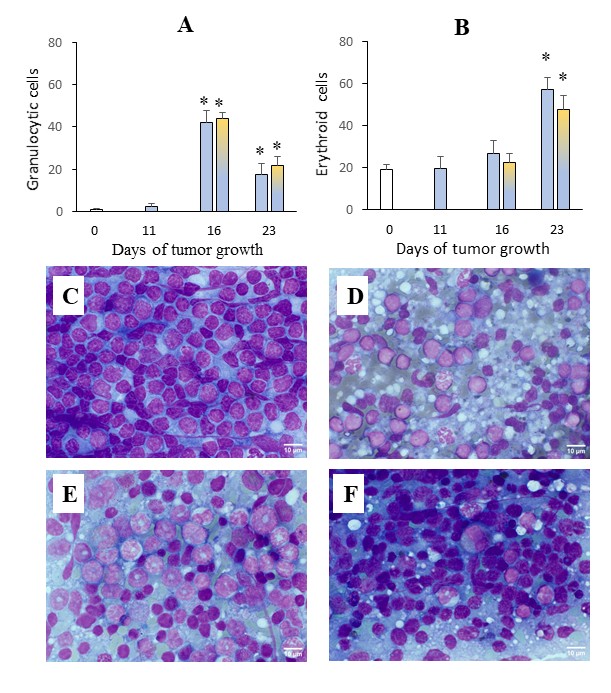
Fig. 7: Effect of UMUS on hematopoiesis in the spleen during CP therapy. Dynamics of granulocytic (A) and erythroid (B) hematopoietic lineages in the spleen during tumor growth after CP administration (blue bars) without UMUS and after UMUS (yellow-blue bars). Spleen imprint smears: intact mouse (C); mouse on day 11 of tumor growth after CP administration (D); mouse on day 16 of tumor growth after CP administration and UMUS (E); mouse on day 23 of tumor growth after CP administration and UMUS (F). * (p<0.05) – statistically significant differences compared with intact mice.
Discussion
Among the possible mechanisms of antitumor therapy, cytotoxic therapy has achieved the greatest development and application over the past 80 years. Its primary goal is to stimulate the death of actively proliferating tumor cells and/or inhibit their proliferative activity. The effectiveness of such therapy is dose-dependent and is largely limited by its high toxicity toward proliferating cells of normal organs and body systems. Therefore, improving treatment outcomes in cancer patients is directly associated with the ability to effectively reduce toxic effects, among which myelotoxicity occupies a leading position. The present study was aimed at developing an effective non-pharmacological method to enhance the regeneration of the hematopoietic system during cytotoxic chemotherapy without reducing its antitumor efficacy. The development of such a method necessarily included an assessment of the relationship between peripheral blood parameters and bone marrow hematopoiesis during cytotoxic therapy with cyclophosphamide (CP), both without and after exposure to ultra-broadband micromechanical ultrasound (UMUS). It should be noted that in clinical practice, the degree of myelosuppression induced by cytotoxic therapy is typically assessed using peripheral blood parameters approximately two weeks after the completion of treatment (i.e., one week before the next treatment cycle). A decrease in these parameters below critical values serves as a basis for initiating supportive antitoxic therapy and/or reducing the dose of antitumor drugs (inevitably leading to reduced efficacy), or even for complete discontinuation of antitumor therapy. The results of our study indicate that such an assessment is not entirely accurate. For example, a dramatic 90% reduction in myelokaryocytes following CP administration and a persistently decreased level (by 40%) two weeks after treatment completion were accompanied by only a ~10% decrease in peripheral blood parameters. Thus, a two-week interval is insufficient for spontaneous regeneration of bone marrow hematopoiesis damaged by cytotoxic agents. This further emphasizes the importance of developing methods to enhance hematopoietic regeneration during cytotoxic chemotherapy without compromising antitumor efficacy, with the potential application of such methods during each treatment cycle, at least for cytostatics with known dose-limiting hematological toxicity. The present study demonstrates that UMUS exerts a pronounced stimulatory effect on the regeneration of specific cellular pools in bone marrow hematopoiesis following CP-induced myelosuppression. A schematic representation of the effects of CP and UMUS on bone marrow hematopoiesis is presented in Fig. 8. CP induces marked suppression of hematopoietic activity, accompanied by depletion of blast cells, differentiated hematopoietic lineages, and myelokaryocytes (Fig. 8A). The most significant effects of bone marrow recovery induced by UMUS were observed in the restoration of the blast cell pool, erythroid lineage cells, and megakaryocytes (Fig. 8C). These results highlight the selective sensitivity of key hematopoietic compartments to mechanophysical stimulation and point to the therapeutic potential of UMUS as an adjunctive modality during cytotoxic therapy. Importantly, UMUS promoted recovery of the hematopoietic system without compromising antitumor efficacy, which is a key requirement for cancer supportive therapy (Fig. 8C). One of the most notable observations was the substantial recovery of blast cells under UMUS exposure. Given that blast cells represent early hematopoietic precursors with high proliferative and differentiation potential, their restoration is critical for effective recovery of bone marrow hematopoiesis. The approximately tenfold reduction in blast cell numbers following CP administration is consistent with previous reports describing the vulnerability of rapidly dividing progenitor cell populations to alkylating agents [20-22]. The subsequent normalization of blast cell numbers under UMUS exposure suggests activation of stem and progenitor cell compartments. This effect is consistent with data indicating that mechanical stimuli can enhance proliferation and self-renewal of hematopoietic stem cells [23]. A significant effect was also observed in the ability of UMUS to stimulate regeneration of the erythroid cell pool, which decreased by approximately 30-fold following CP treatment. Spontaneous regeneration of this cell population two weeks after CP administration resulted in recovery of less than 30% of cells. UMUS significantly stimulated regenerative processes, leading to nearly a twofold increase in erythroid cell numbers. This acceleration of erythropoiesis may be associated with both direct stimulation of erythroid precursors and indirect effects mediated by improved bone marrow microcirculation and stromal support, where niche-dependent signals critically influence erythroid differentiation [24]. In addition, enhanced metabolic activity driven by mechanotransduction may support the biosynthetic demands of erythroid maturation. Recovery of the megakaryocytic lineage, although incomplete, was also significantly improved under UMUS exposure. Megakaryocytes are particularly sensitive to cytotoxic damage due to their large size and complex maturation process. The observed 37-fold decrease following CP administration reflects profound impairment of thrombopoiesis, consistent with previously published data [25-26]. The UMUS-induced doubling of megakaryocyte numbers suggests partial restoration of this lineage, which may be important for correcting thrombocytopenia. Notably, megakaryocyte development is tightly regulated by biomechanical signals within the bone marrow niche, including shear stress and extracellular matrix stiffness [27]. This supports the hypothesis that low-intensity ultrasound may mimic physiological mechanical cues that promote megakaryopoiesis. The observed effects of low-intensity ultrasound on hematopoietic recovery may be explained by mechanotransduction mechanisms. Previous studies have shown that UMUS has been reported to activate signaling pathways such as Wnt/β-catenin, PI3K/Akt, and MAPK, which are critical regulators of proliferation and survival of hematopoietic stem and progenitor cells (Fig. 8B) [28-29]. In this context, UMUS may enhance the regenerative capacity of bone marrow by modulating the stem cell niche and facilitating restoration of hematopoietic homeostasis following cytotoxic injury. Taken together, these results indicate that UMUS may serve as an adjunctive, non-pharmacological complement to cytotoxic chemotherapy, promoting recovery of the hematopoietic system while preserving antitumor efficacy. Further studies are needed to elucidate the underlying molecular mechanisms and to evaluate its potential for clinical application.
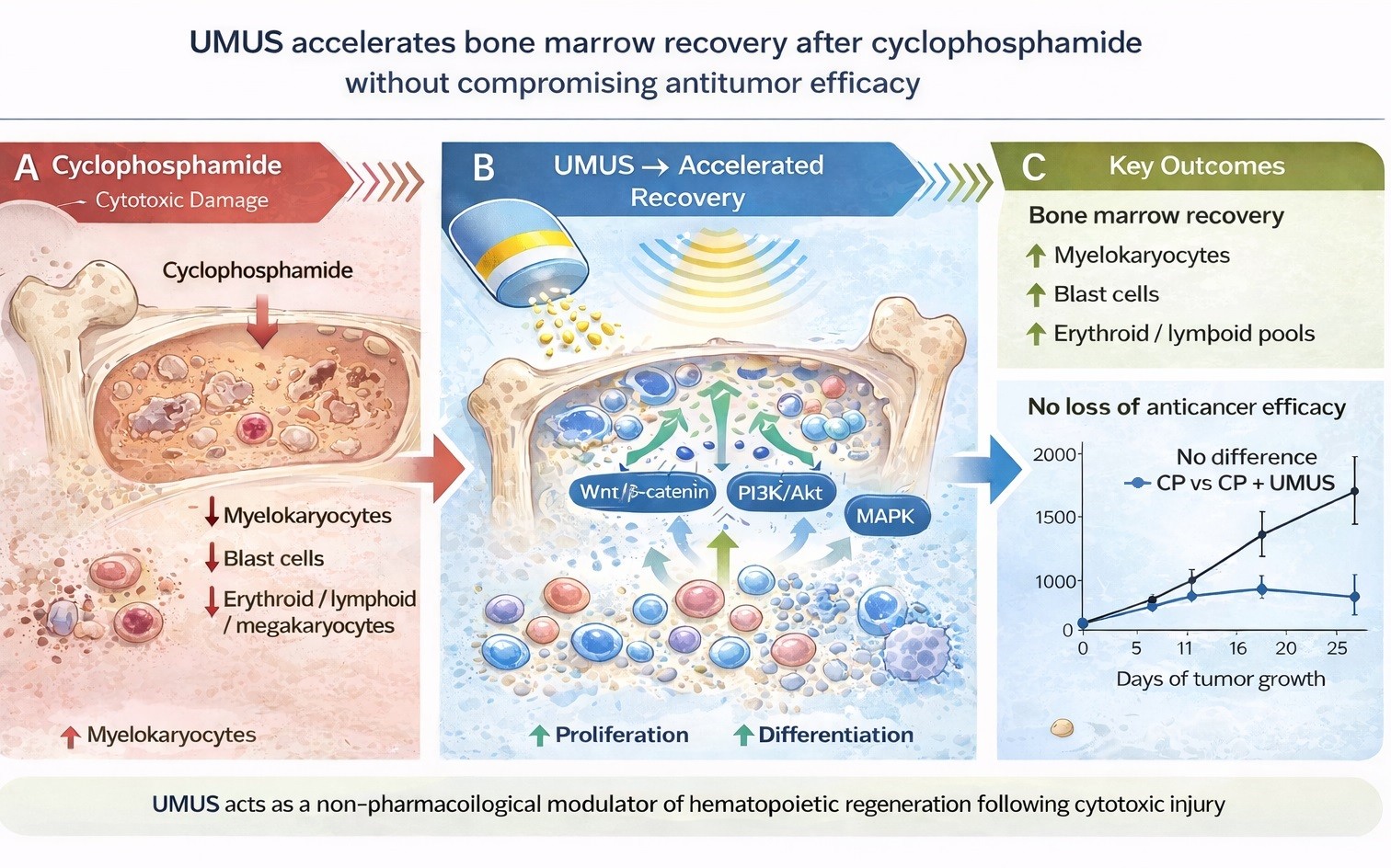
Fig. 8: Schematic representation of the effects of cyclophosphamide (CP) and ultra-broadband micromechanical ultrasound (UMUS) on bone marrow hematopoiesis. CP induces pronounced suppression of hematopoietic activity, characterized by depletion of blast cells, differentiated hematopoietic lineages, and myelokaryocytes (A). Proposed mechanism of UMUS-mediated enhancement of hematopoietic recovery following cyclophosphamide treatment (B). Subsequent exposure to UMUS is associated with accelerated recovery of hematopoiesis without loss of anticancer activity of CP (C).
Conclusion
The results of this study demonstrate the promise of using UMUS as an innovative approach for modifying myelosuppression induced by antitumor cytotoxic drugs without reducing the effectiveness of such therapy. In the future, this opens new prospects for further experimental and clinical studies aimed at integrating mechanotherapeutic strategies into oncological treatment protocols.
Acknowledgements
Funding
This research was sponsored by the Techno-Med Ukraine Project (No. V-01-2025).
Data Availability
The data supporting the findings of experimental results are available from the corresponding author upon reasonable request.
Statement of Ethics
All procedures involving animals were conducted in accordance with institutional guidelines for the care and use of laboratory animals and were approved by the Bioethics Committee of IEPOR NASU (protocol No. 4, dated 04/03/2025).
Author’s contribution
Galina Solyanik: conceptualization, validation, supervision, data curation, writing – original draft, writing – review & editing; Nataliya Rodionova: methodology, investigation, writing – original draft; Yurii V. Stepanov: investigation, writing – original draft; Denis Kolesnik: investigation, validation, visualization; Yulia Yakshibaeva: formal analysis, writing – review & editing; Alexandr Kuzmenko: methodology, investigation, software; Karl Volokh: resources, supervision.
Disclosure Statement
The authors have no conflicts of interest to declare.
References
- Anand U, Dey A, Chandel AKS, Sanyal R, Mishra A, Pandey DK, et al. Cancer chemotherapy and beyond: Current status, drug candidates, associated risks and progress in targeted therapeutics. Genes Dis. 2022;10(4):1367-1401. https://doi.org/10.1016/j.gendis.2022.02.007. Erratum in: Genes Dis. 2024;11(4):101211. https://doi.org/10.1016/j.gendis.2024.101211
- Kurian R, Wang H. Prodrugs in Oncology: Bioactivation and Impact on Therapeutic Efficacy and Toxicity. Int. J. Mol. Sci. 2025;26:988. https://doi.org/10.3390/ijms26030988
- Juthani R, Punatar S, Mittra I. New light on chemotherapy toxicity and its prevention. BJC Rep. 2024;2:41. https://doi.org/10.1038/s44276-024-00064-8
- Maleki EH, Bahrami AR, Matin MM. Cancer cell cycle heterogeneity as a critical determinant of therapeutic resistance. Genes Dis. 2023;11(1):189-204 https://doi.org/10.1016/j.gendis.2022.11.025
- Aboud K, Meissner M, Ocen J, Jones R. Cytotoxic chemotherapy: clinical aspects. Medicine. 2023;51:23-27. https://doi.org/10.1016/j.mpmed.2022.10.005
- Crawford J, Herndon D, Gmitter K, Weiss J. The impact of myelosuppression on quality of life of patients treated with chemotherapy. Future Oncol. 2024;20(21):1515-1530. https://doi.org/10.2217/fon-2023-0513
- Gao A, Zhang L, Zhong D. Chemotherapy-induced thrombocytopenia: literature review. Discov Oncol. 2023;14(1):10. https://doi.org/10.1007/s12672-023-00616-3
- Yang H, Xu X, Tan M, Gao J, Fang R, Liu X, et al. Efficacy and safety of different treatments in chemotherapy-induced thrombocytopenia: a systematic review and network meta-analysis. Front Pharmacol. 2025;16:1549214. https://doi.org/10.3389/fphar.2025.1549214
- Moik F, Ay C. Treatment of VTE in the thrombocytopenic cancer patient. Hematology Am Soc Hematol Educ Program. 2024;2024(1):259-269 https://doi.org/10.1182/hematology.2024000551
- Baig H, Somlo B, Eisen M, Stryker S, Bensink M, Morrow PK. Appropriateness of granulocyte colony-stimulating factor use in patients receiving chemotherapy by febrile neutropenia risk level. J Oncol Pharm Pract. 2019;25(7):1576-1585. https://doi.org/10.1177/1078155218799859
- Campbell K, Chadha N, Dimri S, Wang W, Li E. G-CSF primary prophylaxis use and outcomes in patients receiving chemotherapy at intermediate risk for febrile neutropenia: a scoping review. Expert Rev Hematol. 2022;15(7):619-633. https://doi.org/10.1080/17474086.2022.2093712
- Bohlius J, Bohlke K, Castelli R, Djulbegovic B, Lustberg MB, Martino M, et al. Management of Cancer-Associated Anemia With Erythropoiesis-Stimulating Agents: ASCO/ASH Clinical Practice Guideline Update. J Clin Oncol. 2019;37(15):1336-1351. https://doi.org/10.1200/JCO.18.02142
- Pothacamuri MA, Venugopal A, Chandrashekar N, Banderas Echeverry WM, Appiah JG, AlSalami S, et al. Erythropoiesis-Stimulating Agents (ESAs) in Chronic Kidney Disease and Cancer-Related Anemia: A Narrative Review of Literature. Cureus. 2025;17(9):e93584. https://doi.org/10.7759/cureus.93584
- Castaman G, Pieri L. Management of thrombocytopenia in cancer. Thromb Res. 2018;164(1):S89-S93. https://doi.org/10.1016/j.thromres.2018.02.001
- Yang G, Chen S, Wei M. Unraveling the impact of chemoradiotherapy on hematopoietic function in cancer patients: a metabolomics-driven mechanistic investigation. Mol Cell Biochem. 2026. https://doi.org/10.1007/s11010-026-05488-z
- de Naurois J, Novitzky-Basso I, Gill MJ, Marti Marti F, Cullen MH, Roila F. Management of febrile neutropenia: ESMO Clinical Practice Guidelines, Ann Oncol 2010;21:v252-v256. https://doi.org/10.1093/annonc/mdq196
- Xie S, Jiang X, Wang R, Xie S, Hua Y, Zhou S, Yang Y, Zhang J. Low-intensity pulsed ultrasound promotes the proliferation of human bone mesenchymal stem cells by activating PI3K/AKt signaling pathways. J Cell Biochem. 2019;120(9):15823-15833. https://doi.org/10.1002/jcb.28853
- Tan Y, Guo Y, Reed-Maldonado AB, Li Z, Lin G, Xia SJ, Lue TF. Low-intensity pulsed ultrasound stimulates proliferation of stem/progenitor cells: what we need to know to translate basic science research into clinical applications. Asian J Androl. 2021;23(6):602-610. https://doi.org/10.4103/aja.aja_25_21
- Marchenko A, MiciukeviciusA, inventors; Evolus Tech Ltd, assignee. Device for ultra-wideband micromechanical therapy and method of its operation. United States patent 20240009487A1. 2024 Mar 21.
- Almeer RS, Alnasser M, Aljarba N, AlBasher GI. Effects of Green cardamom (Elettaria cardamomum Maton) and its combination with cyclophosphamide on Ehrlich solid tumors. BMC Complement Med Ther. 2021;21(1):133. https://doi.org/10.1186/s12906-021-03305-2
- Albalawi AE, Althobaiti NA, Alrdahe SS, Alhasani RH, Alaryani FS, BinMowyna MN. Anti-Tumor Effects of Queen Bee Acid (10-Hydroxy-2-Decenoic Acid) Alone and in Combination with Cyclophosphamide and Its Cellular Mechanisms against Ehrlich Solid Tumor in Mice. Molecules. 2021;26(22):7021. https://doi.org/10.3390/molecules26227021
- Zaki HM, Ali KM, Abd Allah MYY, Abouelnaga AM, Abdraboh ME, Hussein O. Metronomic cyclophosphamide and metformin inhibited tumor growth and repopulated tumor-infiltrating lymphocytes in an experimental carcinoma model. BMC Res Notes. 2024;17(1):4. https://doi.org/10.1186/s13104-023-06651-1
- Horton PD, Syed A, Winkler M, Vaidya AB, Rariden M, Arora N, Zhou Y, Kobayashi M, Yoshimoto M, Lee HJ, Kim HE, Hagan JP, Denicourt C, Moore TI, Wenzel PL. Mechanometabolism instructs hematopoietic stem cell specification. J Exp Med. 2026 Mar 2;223(3):e20250607. https://doi.org/10.1084/jem.20250607
- Ward CM, Ravid K. Matrix Mechanosensation in the Erythroid and Megakaryocytic Lineages. Cells. 2020;9(4):894. https://doi.org/10.3390/cells9040894
- Xie RF [Effect of cyclophosphamide on the bone marrow hematopoiesis in the mouse].. Zhonghua Zhong Liu Za Zhi. 1985;7(6):435-437. Chinese.
- Khazaei MR, Kamali H, Khazaei M. Trifolium Pratense Improves Cyclophosphamide-Induced Thrombocytopenia and Leukopenia in a Rat Model of Chemotherapy. Iran J Ped Hematol Oncol. 2024;14(2):94-103. https://doi.org/10.18502/ijpho.v14i2.15268
- Machlus KR, Italiano JE Jr. The incredible journey: From megakaryocyte development to platelet formation. J Cell Biol. 2013;201(6):785-796. https://doi.org/10.1083/jcb.201304054
- Liang C, Liu X, Yan Y, Sun R, Li J, Geng W. Effectiveness and Mechanisms of Low-Intensity Pulsed Ultrasound on Osseointegration of Dental Implants and Biological Functions of Bone Marrow Mesenchymal Stem Cells. Stem Cells Int. 2022;2022:7397335. https://doi.org/10.1155/2022/7397335
- Yijing Han, Hui Gao, Jing Gao, Yonghong Yang, Chengqi He, Low-intensity pulsed ultrasound regulates bone marrow mesenchymal stromal cells differentiation and inhibits bone loss by activating the IL-11-Wnt/β-catenin signaling pathway, Int Immunopharmacol. 2024;143(1):113380. https://doi.org/10.1016/j.intimp.2024.113380